Articles:
2-phenanthrenol, 4b,5,6,7,8,8a,9,10-octahydro-4b,8,8-trimethyl-1-(1-methylethyl)-, (4bS,8aS)-
Notes:
isolated from the bark of podocarpus nagi.
| CAS Number: | 511-15-9 | 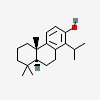 3D/inchi 3D/inchi
|
| FDA UNII: | 67NH2854WW | |
| Nikkaji Web: | J14.012K | |
| MDL: | MFCD02094185 | |
| XlogP3-AA: | 6.70 (est) | |
| Molecular Weight: | 286.45830000 | |
| Formula: | C20 H30 O | |
| NMR Predictor: | Predict (works with chrome or firefox) | |
Category: natural substances and extractives
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
| Google Scholar: | Search |
| Google Books: | Search |
| Google Scholar: with word "volatile" | Search |
| Google Scholar: with word "flavor" | Search |
| Google Scholar: with word "odor" | Search |
| Google Patents: | Search |
| US Patents: | Search |
| EU Patents: | Search |
| Pubchem Patents: | Search |
| PubMed: | Search |
| NCBI: | Search |
Physical Properties:
| Appearance: | white solid (est) |
| Assay: | 97.00 to 100.00 % |
| Food Chemicals Codex Listed: | No |
| Melting Point: | 128.00 to 132.00 °C. @ 760.00 mm Hg |
| Boiling Point: | 369.00 to 371.00 °C. @ 760.00 mm Hg |
| Vapor Pressure: | 0.000005 mmHg @ 25.00 °C. (est) |
| Flash Point: | 332.00 °F. TCC ( 166.67 °C. ) |
| logP (o/w): | 6.405 (est) |
| Soluble in: | |
| alcohol | |
| water, 0.03402 mg/L @ 25 °C (est) | |
| Insoluble in: | |
| water | |
Organoleptic Properties:
| Odor and/or flavor descriptions from others (if found). | |
Cosmetic Information:
| CosIng: | cosmetic data |
| Cosmetic Uses: |
antioxidants deodorants oral care agents |
Suppliers:
| BOC Sciences |
| For experimental / research use only. |
| Totarol >98%
Odor: characteristic Use: Totarol is a naturally produced diterpenoid isolated from the barks of Podocarpus totara. Recent studies show that (+)-totarol has unique antimicrobial and therapeutic properties.
anti-bacteria |
| Charkit Chemical |
| TOTAROL K7 LIQUID |
| Charkit Chemical |
| TOTAROL |
| Santa Cruz Biotechnology |
| For experimental / research use only. |
| (4bS)-trans-8,8-Trimethyl-4b,5,6,7,8,8a,9,10-octahydro-1-isopropylphenanthren-2-ol |
| Sigma-Aldrich: Aldrich |
| For experimental / research use only. |
| 4bS-trans-8,8-Trimethyl-4b,5,6,7,8,8a,9,10-octahydro-1-isopropyl-phenanthren-2-ol 98% |
Safety Information:
| Preferred SDS: View | |
| European information : | |
| Most important hazard(s): | |
| Xi - Irritant | |
|
R 36/37/38 - Irritating to eyes, respiratory system, and skin. S 02 - Keep out of the reach of children. S 24/25 - Avoid contact with skin and eyes. S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S 36 - Wear suitable protective clothing. | |
| Hazards identification | |
| Classification of the substance or mixture | |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) | |
| None found. | |
| GHS Label elements, including precautionary statements | |
| Pictogram | |
| Hazard statement(s) | |
| None found. | |
| Precautionary statement(s) | |
| None found. | |
| Oral/Parenteral Toxicity: | |
| Not determined | |
| Dermal Toxicity: | |
| Not determined | |
| Inhalation Toxicity: | |
| Not determined | |
Safety in Use Information:
| Category: | natural substances and extractives | ||
| Recommendation for totarol usage levels up to: | |||
| not for fragrance use. | |||
| Recommendation for totarol flavor usage levels up to: | |||
| not for flavor use. | |||
Safety References:
| EPI System: | View |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 92783 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WGK Germany: | 3 |
| (4bS,8aS)-4b,8,8-trimethyl-1-propan-2-yl-5,6,7,8a,9,10-hexahydrophenanthren-2-ol | |
| Chemidplus: | 0000511159 |
References:
| (4bS,8aS)-4b,8,8-trimethyl-1-propan-2-yl-5,6,7,8a,9,10-hexahydrophenanthren-2-ol | |
| NIST Chemistry WebBook: | Search Inchi |
| Pubchem (cid): | 92783 |
| Pubchem (sid): | 135049775 |
| Pherobase: | View |
Other Information:
| (IUPAC): | Atomic Weights of the Elements 2011 (pdf) |
| Videos: | The Periodic Table of Videos |
| tgsc: | Atomic Weights use for this web site |
| (IUPAC): | Periodic Table of the Elements |
| CHEBI: | View |
| CHEMBL: | View |
| HMDB (The Human Metabolome Database): | Search |
| Typical G.C. | |
| VCF-Online: | VCF Volatile Compounds in Food |
| ChemSpider: | View |
| Wikipedia: | View |
Potential Blenders and core components note
| None Found | ||
Potential Uses:
| None Found |
Occurrence (nature, food, other): note
| cedar western red cedar Search Trop Picture | |
| cedarleaf oil canada @ trace-0.10% Data GC Search Trop Picture | |
| ferula communis Search Trop Picture | |
| juniperus procera Search Trop Picture | |
| plumbago zeylanica Search Trop Picture | |
| podocarpus nagi Search Trop Picture | |
| podocorpus totara g. benn. Search Trop Picture | |
| rosemary oil CO2 extract @ trace% Data GC Search Trop Picture |
Synonyms:
| 4b,5,6,7,8,8a,9,10- | octahydro-4b,8,8-trimethyl-1-(1-methyl ethyl)-2-phenanthrenol |
| 4b,5,6,7,8,8a,9,10- | octahydro-4b,8,8-trimethyl-1-(1-methylethyl)-2-phenanthrenol |
| 2- | phenanthrenol, 4b,5,6,7,8,8a,9,10-octahydro-4b,8,8-trimethyl-1-(1-methylethyl)-, (4bS-trans)- |
| 2- | phenanthrenol, 4b,5,6,7,8,8a,9,10-octahydro-4b,8,8-trimethyl-1-(1-methylethyl)-, (4bS,8aS)- |
| 14-iso | propyl podocarpa-8,11,13-trien-13-ol |
| (4bS,8aS)-4b,8,8- | trimethyl-1-(propan-2-yl)-4b,5,6,7,8,8a,9,10-octahydrophenanthren-2-ol |
| (4bS,8aS)-4b,8,8- | trimethyl-1-propan-2-yl-5,6,7,8a,9,10-hexahydrophenanthren-2-ol |
| 4bS-trans-8,8- | trimethyl-4b,5,6,7,8,8a,9,10-octahydro-1-isopropyl-phenanthren-2-ol |
| (4bS)-trans-8,8- | trimethyl-4b,5,6,7,8,8a,9,10-octahydro-1-isopropylphenanthren-2-ol |

