Articles:
prenylthiol
Notes:
Substance responsible for ``sun-struck'' flavour of beer. Found in coffee. Used in food flavourings
| CAS Number: | 5287-45-6 | 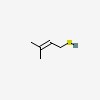 3D/inchi 3D/inchi
|
| FDA UNII: | FDG262156U | |
| Nikkaji Web: | J534.118C | |
| MDL: | MFCD00068639 | |
| CoE Number: | 11511 | |
| XlogP3-AA: | 2.00 (est) | |
| Molecular Weight: | 102.19970000 | |
| Formula: | C5 H10 S | |
| NMR Predictor: | Predict (works with chrome, Edge or firefox) | |
Category: flavoring agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
| Google Scholar: | Search |
| Google Books: | Search |
| Google Scholar: with word "volatile" | Search |
| Google Scholar: with word "flavor" | Search |
| Google Scholar: with word "odor" | Search |
| Perfumer and Flavorist: | Search |
| Google Patents: | Search |
| US Patents: | Search |
| EU Patents: | Search |
| Pubchem Patents: | Search |
| PubMed: | Search |
| NCBI: | Search |
| JECFA Food Flavoring: | 522 prenylthiol |
| DG SANTE Food Flavourings: | 12.170 3-methylbut-2-ene-1-thiol |
| FEMA Number: | 3896 prenylthiol |
| FDA: | No longer provide for the use of these seven synthetic flavoring substances |
| FDA Mainterm (SATF): | 5287-45-6 ; PRENYLTHIOL |
Physical Properties:
Organoleptic Properties:
| Odor Type: sulfurous | |
| sulfurous smoky leek onion skunk | |
| Odor Description: at 0.10 % in propylene glycol. | sulfurous smoke leek onion skunky |
| Odor and/or flavor descriptions from others (if found). | |
| R C Treatt & Co Ltd | |
| Prenyl mercaptan 1% Triacetin Kosher | |
| Odor Description: | Intense leek, onion-like, skunky |
Cosmetic Information:
| None found |
Suppliers:
Safety Information:
| Preferred SDS: View | |
| European information : | |
| Most important hazard(s): | |
| Xi - Irritant | |
|
R 10 - Flammable. R 36/37/38 - Irritating to eyes, respiratory system, and skin. S 02 - Keep out of the reach of children. S 16 - Keep away from sources of ignition - No Smoking. S 24/25 - Avoid contact with skin and eyes. S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S 36/37/39 - Wear suitable clothing, gloves and eye/face protection. | |
| Hazards identification | |
| Classification of the substance or mixture | |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) | |
| None found. | |
| GHS Label elements, including precautionary statements | |
| Pictogram | |
| Hazard statement(s) | |
| None found. | |
| Precautionary statement(s) | |
| None found. | |
| Oral/Parenteral Toxicity: | |
| Not determined | |
| Dermal Toxicity: | |
| Not determined | |
| Inhalation Toxicity: | |
| Not determined | |
Safety in Use Information:
| Category: | flavoring agents | ||
| Recommendation for prenyl mercaptan usage levels up to: | |||
| not for fragrance use. | |||
| Maximised Survey-derived Daily Intakes (MSDI-EU): | ND (μg/capita/day) | ||
| Structure Class: | I | ||
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). | |||
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library | |||
| publication number: 18 | |||
| Click here to view publication 18 | |||
| average usual ppm | average maximum ppm | ||
| baked goods: | 0.05000 | 5.00000 | |
| beverages(nonalcoholic): | 0.00100 | 5.00000 | |
| beverages(alcoholic): | 0.00500 | 0.05000 | |
| breakfast cereal: | 0.01000 | 5.00000 | |
| cheese: | - | - | |
| chewing gum: | 0.05000 | 0.10000 | |
| condiments / relishes: | - | - | |
| confectionery froastings: | 0.01000 | 0.05000 | |
| egg products: | - | - | |
| fats / oils: | - | - | |
| fish products: | - | - | |
| frozen dairy: | 0.10000 | 5.00000 | |
| fruit ices: | - | - | |
| gelatins / puddings: | - | - | |
| granulated sugar: | - | - | |
| gravies: | 0.10000 | 5.00000 | |
| hard candy: | 0.01000 | 0.05000 | |
| imitation dairy: | - | - | |
| instant coffee / tea: | 0.00100 | 0.01000 | |
| jams / jellies: | - | - | |
| meat products: | 0.01000 | 5.00000 | |
| milk products: | 0.00500 | 5.00000 | |
| nut products: | - | - | |
| other grains: | - | - | |
| poultry: | - | - | |
| processed fruits: | - | - | |
| processed vegetables: | - | - | |
| reconstituted vegetables: | - | - | |
| seasonings / flavors: | 0.20000 | 2.00000 | |
| snack foods: | - | - | |
| soft candy: | 0.10000 | 5.00000 | |
| soups: | 0.10000 | 5.00000 | |
| sugar substitutes: | - | - | |
| sweet sauces: | 0.10000 | 5.00000 | |
Safety References:
| European Food Safety Athority(EFSA): | Flavor usage levels; Subacute, Subchronic, Chronic and Carcinogenicity Studies; Developmental / Reproductive Toxicity Studies; Genotoxicity Studies... |
| European Food Safety Authority (EFSA) reference(s): | |
| Flavouring Group Evaluation 8 (FGE.08)[1]: Aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups from chemical group 20 View page or View pdf | |
| Flavouring Group Evaluation 8, Revision 1 (FGE.08Rev1): Aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups from chemical groups 20 and 30 View page or View pdf | |
| Scientific Opinion on Flavouring Group Evaluation 8, Revision 3 (FGE.08Rev3): Aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups from chemical groups 20 and 30 View page or View pdf | |
| Scientific Opinion on Flavouring Group Evaluation 08, Revision 4 (FGE.08Rev4): Aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups from chemical groups 20 and 30 View page or View pdf | |
| EPI System: | View |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 146586 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WISER: | UN 3336 |
| WGK Germany: | 3 |
| 3-methylbut-2-ene-1-thiol | |
| Chemidplus: | 0005287456 |
References:
| 3-methylbut-2-ene-1-thiol | |
| NIST Chemistry WebBook: | Search Inchi |
| Pubchem (cid): | 146586 |
| Pubchem (sid): | 135103905 |
| Flavornet: | 5287-45-6 |
Other Information:
| (IUPAC): | Atomic Weights of the Elements 2011 (pdf) |
| Videos: | The Periodic Table of Videos |
| tgsc: | Atomic Weights use for this web site |
| (IUPAC): | Periodic Table of the Elements |
| FDA Substances Added to Food (formerly EAFUS): | View |
| HMDB (The Human Metabolome Database): | HMDB31529 |
| FooDB: | FDB008136 |
| YMDB (Yeast Metabolome Database): | YMDB01604 |
| Export Tariff Code: | 2930.90.8599 |
| VCF-Online: | VCF Volatile Compounds in Food |
| ChemSpider: | View |
| FAO: | Prenylthiol |
Potential Blenders and core components note
Potential Uses:
| coffee dark roast coffee | FL | |
| fish | FL | |
| garlic roasted garlic | FL | |
| meat | FL | |
| meat grilled meat | FL |
Occurrence (nature, food, other): note
| beer Search PMC Picture | |
| coffee Search PMC Picture | |
| hop oil Search Trop Picture |
Synonyms:
| but-2-ene-1-thiol, 3-methyl- | |
| 2- | butene-1-thiol, 3-methyl- |
| 3- | methyl but-2-ene-1-thiol |
| methyl butene thiol | |
| 3- | methyl-2-buten-1-thiol |
| 3- | methyl-2-butene-1-thiol |
| 3- | methyl-2-butenyl mercaptan |
| 3- | methylbut-2-ene-1-thiol |
| prenyl mercaptan 1% in triacetin | |
| prenyl mercaptan natural 1% in triacetin | |
| prenyl thiol | |
| prenylthiol |











