|
Category: flavor and fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
Physical Properties:
| Appearance: | colorless clear liquid (est) |
| Assay: | 99.00 to 100.00 %
|
| Food Chemicals Codex Listed: | No |
| Specific Gravity: | 0.84400 to 0.85200 @ 25.00 °C.
|
| Pounds per Gallon - (est).: | 7.023 to 7.089
|
| Refractive Index: | 1.43800 to 1.44800 @ 20.00 °C.
|
| Boiling Point: | 138.00 to 174.00 °C. @ 760.00 mm Hg
|
| Vapor Pressure: | 2.384000 mmHg @ 25.00 °C. (est) |
| Flash Point: | 110.00 °F. TCC ( 43.33 °C. )
|
| logP (o/w): | 1.055 (est) |
| Soluble in: |
| | alcohol | | | water, 4.094e+004 mg/L @ 25 °C (est) |
Organoleptic Properties:
| |
| Odor Type: fruity |
| |
| | fruity green lavender fermented yeasty |
Odor Description:
at 100.00 %. | fruity green lavender |
| |
| | sweet fruity alcoholic green |
Odor Description:
| Sweet, fruity, alcoholic with a green nuance
Mosciano, Gerard P&F 16, No. 4, 45, (1991) |
| |
| |
| Flavor Type: fruity |
| |
| | green fruity |
Taste Description:
at 30.00 ppm. | Green, fruity
Mosciano, Gerard P&F 16, No. 4, 45, (1991) |
| |
| Odor and/or flavor descriptions from others (if found). |
| |
| Bedoukian Research |
| PRENOL (3-METHYL-2-BUTEN-1-OL) ≥96.0%, Kosher |
| Odor Description: | fruity, green, lavender
Used in lavender and hops compositions. |
| Taste Description: | green fruity
Works particularly well in passion fruit and cranberry flavors. |
| |
| Moellhausen |
| PRENOL |
| Odor Description: | fresh, ethereal, yeast and fermented yeasty |
| |
| Indukern F&F |
| PRENOL |
| Odor Description: | FRUITY, GREEN, LAVENDER |
| |
| |
Cosmetic Information:
Suppliers:
| Beijing Lys Chemicals |
| 3-Methyl-2-buten-1-ol
|
| Berjé |
| Prenol
|
| Media |
| BOC Sciences |
| For experimental / research use only. |
| PRENOL 98.0%
|
| Citrus and Allied Essences |
| Prenol
|
| Market Report |
| Ernesto Ventós |
| PRENOL
Odor: HERBAL,GREEN,FRUITY,LAVENDER Flavor: FRESH,FRUITY,GREEN,APPLE |
| Grau Aromatics |
| VERTENOL (PRENOL)
NI, Kosher |
| Indukern F&F |
| PRENOL
Odor: FRUITY, GREEN, LAVENDER |
| Lluch Essence |
| PRENOL
|
| M&U International |
| Prenol
|
| Moellhausen |
| PRENOL
Odor: fresh, ethereal, yeast and fermented yeasty |
| Penta International |
| 3-METHYL-2-BUTEN-1-OL
|
| Santa Cruz Biotechnology |
| For experimental / research use only. |
| 3-Methyl-2-buten-1-ol
|
| Sigma-Aldrich |
| 3-Methyl-2-buten-1-ol, ≥98%, FG
Odor: fruity; green |
| Certified Food Grade Products |
| SRS Aromatics |
| VERTENOL
|
| Synerzine |
| 3-Methyl-2-buten-1-ol
|
| TCI AMERICA |
| For experimental / research use only. |
| 3-Methyl-2-buten-1-ol >98.0%(GC)
|
| United International |
| Prenol
|
Safety Information:
| Preferred SDS: View |
| European information : |
| Most important hazard(s): | | Xn - Harmful. |
R 10 - Flammable.
R 22 - Harmful if swallowed.
R 36/38 - Irritating to skin and eyes.
S 02 - Keep out of the reach of children.
S 16 - Keep away from sources of ignition - No Smoking.
S 20/21 - When using do not eat, drink or smoke.
S 24/25 - Avoid contact with skin and eyes.
S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S 37/39 - Wear suitable gloves and eye/face protection.
|
| |
| Hazards identification |
| |
| Classification of the substance or mixture |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) |
| None found. |
| GHS Label elements, including precautionary statements |
| |
| Pictogram | |
| |
| Hazard statement(s) |
| None found. |
| Precautionary statement(s) |
| None found. |
| Oral/Parenteral Toxicity: |
oral-rat LD50 810 mg/kg
Food and Cosmetics Toxicology. Vol. 17, Pg. 895, 1979.
|
| Dermal Toxicity: |
skin-rabbit LD50 3900 mg/kg
Food and Cosmetics Toxicology. Vol. 17, Pg. 895, 1979.
|
| Inhalation Toxicity: |
|
Not determined
|
Safety in Use Information:
| Category: | flavor and fragrance agents |
| RIFM Fragrance Material Safety Assessment: Search |
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice |
| Recommendation for prenol usage levels up to: | | | 8.0000 % in the fragrance concentrate.
|
| |
| Maximised Survey-derived Daily Intakes (MSDI-EU): | 4.60 (μg/capita/day) |
| Maximised Survey-derived Daily Intakes (MSDI-USA): | 3.80 (μg/capita/day) |
| Threshold of Concern: | 1800 (μg/person/day) |
| Structure Class: | I |
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). |
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library |
| publication number: 12 |
| Click here to view publication 12 |
| | average usual ppm | average maximum ppm |
| baked goods: | - | 3.00000 |
| beverages(nonalcoholic): | - | 1.00000 |
| beverages(alcoholic): | - | - |
| breakfast cereal: | - | - |
| cheese: | - | - |
| chewing gum: | - | 20.00000 |
| condiments / relishes: | - | - |
| confectionery froastings: | - | 2.00000 |
| egg products: | - | - |
| fats / oils: | - | - |
| fish products: | - | - |
| frozen dairy: | - | - |
| fruit ices: | - | 1.50000 |
| gelatins / puddings: | - | 2.00000 |
| granulated sugar: | - | - |
| gravies: | - | - |
| hard candy: | - | 5.00000 |
| imitation dairy: | - | - |
| instant coffee / tea: | - | - |
| jams / jellies: | - | 2.00000 |
| meat products: | - | - |
| milk products: | - | - |
| nut products: | - | - |
| other grains: | - | - |
| poultry: | - | - |
| processed fruits: | - | - |
| processed vegetables: | - | - |
| reconstituted vegetables: | - | - |
| seasonings / flavors: | - | - |
| snack foods: | - | - |
| soft candy: | - | 2.50000 |
| soups: | - | - |
| sugar substitutes: | - | - |
| sweet sauces: | - | - |
Safety References:
| European Food Safety Athority(EFSA): | Flavor usage levels; Subacute, Subchronic, Chronic and Carcinogenicity Studies; Developmental / Reproductive Toxicity Studies; Genotoxicity Studies... |
| European Food Safety Authority (EFSA) reference(s): |
Flavouring Group Evaluation 202: 3-Alkylated aliphatic acyclic alpha,beta-unsaturated aldehydes and precursors with or without additional double bonds from chemical subgroup 1.1.3 of FGE.19[1]
View page or View pdf |
Scientific Opinion on Flavouring Group Evaluation 20, Revision 2 (FGE.20Rev2): Benzyl alcohols, benzaldehydes, a related acetal, benzoic acids, and related esters from chemical groups 23 and 30
View page or View pdf |
Flavouring Group Evaluation 72 (FGE.72): Consideration of aliphatic, branched-chain saturated and unsaturated alcohols, aldehydes, acids, and related esters evaluated by the JECFA (61st meeting) structurally related to branched- and straight-chain unsaturated carboxylic acids. Esters of these and straight-chain aliphatic saturated alcohols evaluated by EFSA in FGE.05Rev2 (2010)
View page or View pdf |
Scientific Opinion on Flavouring Group Evaluation 20, Revision 3(FGE.20Rev3): Benzyl alcohols, benzaldehydes, a related acetal, benzoic acids, and related esters from chemical groups 23 and 30
View page or View pdf |
Scientific Opinion on Flavouring Group Evaluation 72, Revision 1 (FGE.72Rev1): Consideration of aliphatic, branched-chain saturated and unsaturated alcohols, aldehydes, acids, and related esters evaluated by the JECFA (61st meeting) structurally related to branched- and straight-chain unsaturated carboxylic acids, esters of these and straight-chain aliphatic saturated alcohols evaluated by EFSA in FGE.05Rev2
View page or View pdf |
Scientific Opinion on Flavouring Group Evaluation 72, Revision 2 (FGE.72Rev2): consideration of aliphatic, branched-chain saturated and unsaturated alcohols, aldehydes, acids and related esters evaluated by JECFA (61st, 68th and 69th meetings) and structurally related to flavouring substances in FGE.05Rev3
View page or View pdf |
| EPI System: | View |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA Substance Registry Services (TSCA): | 556-82-1 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 11173 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WISER: | UN 1987 |
| WGK Germany: | 1 |
| | 3-methylbut-2-en-1-ol |
| Chemidplus: | 0000556821 |
| RTECS: | EM9472500 for cas# 556-82-1 |
References:
Other Information:
Potential Blenders and core components note
| |
| For Odor |
| No odor group found for these |
| 3- | mercapto-3-methyl-1-hexanol | |
| 4-(1- | propenyl) pyridine | |
| | satinaldehyde | FL/FR |
| 1-( | thienyl-2)butan-1,2-dione | |
|
| (E)- | tiglaldehyde | FL/FR |
| animal |
| para- | cresyl isobutyrate | FL/FR |
| | methyl (E)-2-octenoate | FL/FR |
| balsamic |
| iso | amyl benzoate | FL/FR |
| | hexyl benzoate | FL/FR |
| brown |
| sec- | heptyl acetate | FL/FR |
| chemical |
| | propyl propionate | FL/FR |
| citrus |
| 2- | heptanol | FL/FR |
| creamy |
| 3- | heptyl dihydro-5-methyl-2(3H)-furanone | FL/FR |
| earthy |
| | methyl 3-hexenoate | FL/FR |
| ethereal |
| | acetal | FL/FR |
| | acetaldehyde dimethyl acetal | FL/FR |
| | cyclohexyl formate | FL/FR |
| 2,6- | dimethyl-4-heptanol | FL/FR |
| 1- | hexen-3-ol | FL/FR |
| | methyl ethyl ketone | FL/FR |
| 2- | methyl valeraldehyde | FL/FR |
| | propyl formate | FL/FR |
| fatty |
| 3- | decen-2-one | FL/FR |
| | ethyl undecylenate | FL/FR |
| | hexyl pivalate | FR |
| | methyl (E)-2-hexenoate | FL/FR |
| | methyl 2-hexenoate | FL/FR |
| fermented |
| 3- | methyl-1-pentanol | FL/FR |
| | propyl nonanoate | FL/FR |
| floral |
| alpha- | amyl cinnamaldehyde diethyl acetal | FR |
| | boronia absolute | FL/FR |
| | citronellol | FL/FR |
| | citronellyl acetate | FL/FR |
| | citronellyl formate | FL/FR |
| | citronellyl propionate | FL/FR |
| | cuminyl acetaldehyde | FL/FR |
| beta- | damascenone | FL/FR |
| gamma- | damascone | FR |
| 6,8- | dimethyl-2-nonanol | FR |
| | ethyl linalool | FR |
| | geranyl acetate | FL/FR |
| (E)- | geranyl acetone | FL/FR |
| beta- | ionone | FL/FR |
| iso | jasmone | FL/FR |
| iso | jasmone | FL/FR |
| | lilac pentanol | FL/FR |
| | lily propanol | FR |
| (Z)- | methyl epi-jasmonate | FL/FR |
| | methyl jasmonate | FL/FR |
| | nerol | FL/FR |
| | neryl formate | FL/FR |
| | papaya isobutyrate | FL/FR |
| | phenethyl acetate | FL/FR |
| 2- | phenyl propionaldehyde dimethyl acetal | FL/FR |
| | styralyl propionate | FL/FR |
| fruity |
| | acetaldehyde dihexyl acetal | FL/FR |
| | acetaldehyde diisoamyl acetal | FL/FR |
| | allyl 2-ethyl butyrate | FL/FR |
| | allyl amyl glycolate | FR |
| | allyl butyrate | FL/FR |
| iso | amyl butyrate | FL/FR |
| | amyl formate | FL/FR |
| iso | amyl hexanoate | FL/FR |
| | amyl hexanoate | FL/FR |
| iso | amyl isobutyrate | FL/FR |
| iso | amyl isovalerate | FL/FR |
| iso | amyl octanoate | FL/FR |
| | berry pentadienoate | FL/FR |
| | bisabolene | FL/FR |
| | butyl 2-decenoate | FL/FR |
| | butyl 2-methyl butyrate | FL/FR |
| | butyl hexanoate | FL/FR |
| | butyl isobutyrate | FL/FR |
| iso | butyl isovalerate | FL/FR |
| iso | butyl propionate | FL/FR |
| | cherry pentenoate | FL/FR |
| | citronellyl isobutyrate | FL/FR |
| 2- | cyclopentyl cyclopentanone | FL/FR |
| (E)-beta- | damascone | FL/FR |
| (Z)-beta- | damascone | FL/FR |
| (E)-alpha- | damascone | FL/FR |
| | diethyl malonate | FL/FR |
| | dimethyl succinate | FL/FR |
| | ethyl (E)-3-hexenoate | FL/FR |
| | ethyl 2-cyclohexyl propionate | FR |
| | ethyl 2-octenoate | FL/FR |
| | ethyl 3-acetoxyhexanoate | FL/FR |
| | ethyl 3-hexenoate | FL/FR |
| | ethyl 3,5,5-trimethyl hexanoate | FR |
| | ethyl acetoacetate | FL/FR |
| | ethyl levulinate | FL/FR |
| | ethyl methyl-para-tolyl glycidate | FL/FR |
| (E)- | ethyl tiglate | FL/FR |
| | geranyl butyrate | FL/FR |
| | geranyl isovalerate | FL/FR |
| | grape butyrate | FL/FR |
| | heptanal cyclic ethylene acetal | FR |
| | heptyl butyrate | FL/FR |
| | heptyl isobutyrate | FL/FR |
| | hexanal propylene glycol acetal | FL/FR |
| 2- | hexen-1-ol | FL/FR |
| (Z)-3- | hexen-1-yl 2-methyl-2-pentenoate | FR |
| (E)-3- | hexen-1-yl acetate | FL/FR |
| (Z)-3- | hexen-1-yl isobutyrate | FL/FR |
| | hexyl (E)-tiglate | FL/FR |
| | hexyl acetate | FL/FR |
| | hexyl isovalerate | FL/FR |
| | hexyl propionate | FL/FR |
| | linalyl hexanoate | FL/FR |
| | methyl 3-nonenoate | FL/FR |
| | methyl heptanoate | FL/FR |
| | methyl valerate | FL/FR |
| 3- | methyl-2-butenal | FL/FR |
| | nerolidyl isobutyrate | FR |
| | neryl propionate | FL/FR |
| 2- | nonanone | FL/FR |
| | octen-1-yl cyclopentanone | FL/FR |
| | octyl butyrate | FL/FR |
| 2- | pentyl furan | FL/FR |
| 4- | phenyl-2-butyl acetate | FL/FR |
| | pineapple pentenoate | FL/FR |
| | prenyl hexanoate | FL/FR |
| iso | propyl 2-methyl butyrate | FL/FR |
| | rhubarb undecane | FR |
| | tetrahydrofurfuryl butyrate | FL/FR |
| | tropical indene | FR |
| | tropical thiazole | FL/FR |
| green |
| | acetaldehyde benzyl 2-methoxyethyl acetal | FL/FR |
| | acetaldehyde methyl hexyl acetal | FR |
| | actinidia chinensis fruit extract | FL/FR |
| | butyl heptanoate | FL/FR |
| | cognac heptanone | FL/FR |
| alpha- | decalactone | FL/FR |
| | ethyl (E)-2-hexenoate | FL/FR |
| | ethyl (E)-4-decenoate | FL/FR |
| | green dioxolane | FR |
| (E)-3- | hexen-1-ol | FL/FR |
| (Z)-3- | hexen-1-yl (E)-2-hexenoate | FL/FR |
| (Z)-3- | hexen-1-yl (Z)-3-hexenoate | FL/FR |
| (E)-2- | hexen-1-yl acetate | FL/FR |
| (Z)-3- | hexen-1-yl acetate | FL/FR |
| (Z)-3- | hexen-1-yl benzoate | FL/FR |
| (E)-2- | hexen-1-yl formate | FL/FR |
| (E)-2- | hexen-1-yl hexanoate | FL/FR |
| (Z)-3- | hexen-1-yl hexanoate | FL/FR |
| (Z)-3- | hexen-1-yl isovalerate | FL/FR |
| (Z)-3- | hexen-1-yl octanoate | FL/FR |
| (E)-2- | hexen-1-yl propionate | FL/FR |
| (E)-2- | hexen-1-yl valerate | FL/FR |
| (Z)-3- | hexen-1-yl valerate | FL/FR |
| 3- | hexenyl 2-methyl butyrate | FL/FR |
| 2- | hexenyl acetate | FL/FR |
| (Z)-3- | hexenyl methyl ether | FR |
| | hexyl butyrate | FL/FR |
| | hexyl hexanoate | FL/FR |
| | hexyl isobutyrate | FL/FR |
| | hexyl octanoate | FL/FR |
| | manzanate (Givaudan) | FL/FR |
| | methyl (E)-3-hexenoate | FL/FR |
| | methyl octine carbonate | FL/FR |
| | methyl R-3-acetoxyhexanoate | |
| | neryl butyrate | FL/FR |
| 3- | octyl formate | FL/FR |
| (E)-2- | pentenal | FL/FR |
| | phenoxyethyl isobutyrate | FL/FR |
| 1- | phenyl-2-pentanol | FL/FR |
| alpha-iso | propyl phenyl acetaldehyde | FL/FR |
| | propylene acetal | FL/FR |
| | sorbyl isobutyrate | FL/FR |
| | terpinyl propionate | FL/FR |
| | thiogeraniol | FL/FR |
| | tiglaldehyde | FL/FR |
| | violet dienyne | FR |
| herbal |
| | hexanol | FL/FR |
| melon |
| 2- | octenyl acetate | FL/FR |
| sulfurous |
| | buchu mercaptan | FL/FR |
| terpenic |
| | cassis bud oil | FL/FR |
| tropical |
| beta- | cyclocitral | FL/FR |
| | psidium guajava fruit extract | FL/FR |
| waxy |
| 9- | decenoic acid | FL/FR |
| (E,E)-2,4- | dodecadien-1-ol | |
| (E)- | methyl geranate | FL/FR |
| | methyl octanoate | FL/FR |
| 2- | nonanol | FL/FR |
| | octyl 2-methyl butyrate | FL/FR |
| woody |
| | verdoxan | FR |
| | woody acetate | FR |
| |
| For Flavor |
| |
| No flavor group found for these |
| | acetaldehyde benzyl 2-methoxyethyl acetal | FL/FR |
| | acetaldehyde diisoamyl acetal | FL/FR |
| | allyl tiglate | FL |
| 2- | cyclopentyl cyclopentanone | FL/FR |
| alpha- | decalactone | FL/FR |
| 2,6- | dimethyl-4-heptanol | FL/FR |
| (E,E)-2,4- | dodecadien-1-ol | |
| | ethyl 3-acetoxyhexanoate | FL/FR |
| (E,E)-2,4- | heptadien-1-ol | FL |
| 2- | hexenal | FL |
| 2- | hexenal diethyl acetal | FL |
| 2- | hexenyl acetate | FL/FR |
| | hexyl (E)-2-hexenoate | FL |
| | hexyl propionate | FL/FR |
| | linalyl hexanoate | FL/FR |
| | methyl (E)-2-hexenoate | FL/FR |
| | methyl 2-hexenoate | FL/FR |
| | methyl 4-pentenoate | FL |
| (E)- | methyl geranate | FL/FR |
| 2- | methyl-2-octenal | FL |
| (E)-2- | methyl-2-octenal | FL |
| 3- | methyl-3-pentanol | FL |
| (E,E)-3,5- | octadien-2-one | FL |
| 4- | phenyl-2-butyl acetate | FL/FR |
| | prenyl hexanoate | FL/FR |
| | propyl nonanoate | FL/FR |
| alpha-iso | propyl phenyl acetaldehyde | FL/FR |
| | propylene acetal | FL/FR |
| (E)- | tiglaldehyde | FL/FR |
|
| | sorbyl isobutyrate | FL/FR |
| alliaceous |
| | tropical thiazole | FL/FR |
| apple |
| green | apple concentrate | FL |
| aromatic |
| para- | cresyl isobutyrate | FL/FR |
| astringent |
| 1-( | thienyl-2)butan-1,2-dione | |
| berry |
| | heptyl isobutyrate | FL/FR |
| cheesy |
| 2- | nonanone | FL/FR |
| chemical |
| | methyl ethyl ketone | FL/FR |
| citrus |
| | bisabolene | FL/FR |
| | cognac heptanone | FL/FR |
| | nerol | FL/FR |
| | styralyl propionate | FL/FR |
| coconut |
| (R)- | massoia lactone | FL |
| cooling |
| | manzanate (Givaudan) | FL/FR |
| creamy |
| | massoia lactone | FL |
| earthy |
| 1- | hexen-3-yl acetate | FL |
| estery |
| | ethyl acetoacetate | FL/FR |
| ethereal |
| | acetaldehyde dimethyl acetal | FL/FR |
| | allyl 2-ethyl butyrate | FL/FR |
| fatty |
| | ethyl (E)-4-decenoate | FL/FR |
| | ethyl undecylenate | FL/FR |
| sec- | heptyl acetate | FL/FR |
| (Z)-3- | hexen-1-yl benzoate | FL/FR |
| floral |
| | citronellol | FL/FR |
| | citronellyl acetate | FL/FR |
| | citronellyl propionate | FL/FR |
| (E)- | geranyl acetone | FL/FR |
| (Z)- | methyl epi-jasmonate | FL/FR |
| | methyl jasmonate | FL/FR |
| | satinaldehyde | FL/FR |
| fruity |
| iso | amyl benzoate | FL/FR |
| | amyl formate | FL/FR |
| iso | amyl hexanoate | FL/FR |
| | amyl hexanoate | FL/FR |
| iso | amyl isobutyrate | FL/FR |
| iso | amyl octanoate | FL/FR |
| green | apple flavor | FL |
| | apple grape flavor | FL |
| | berry pentadienoate | FL/FR |
| | boronia absolute | FL/FR |
| | butyl 2-decenoate | FL/FR |
| | butyl 2-methyl butyrate | FL/FR |
| | butyl heptanoate | FL/FR |
| | butyl hexanoate | FL/FR |
| | butyl isobutyrate | FL/FR |
| iso | butyl propionate | FL/FR |
| | cassis bud oil | FL/FR |
| | cherry pentenoate | FL/FR |
| | citronellyl formate | FL/FR |
| | citronellyl isobutyrate | FL/FR |
| (E)-beta- | damascone | FL/FR |
| (E)-alpha- | damascone | FL/FR |
| (Z)-beta- | damascone | FL/FR |
| | diethyl malonate | FL/FR |
| | dimethyl succinate | FL/FR |
| | ethyl (E)-2-hexenoate | FL/FR |
| | ethyl (E)-2-octenoate | FL |
| | ethyl (E)-3-hexenoate | FL/FR |
| | ethyl 2-octenoate | FL/FR |
| | ethyl 3-hexenoate | FL/FR |
| | ethyl levulinate | FL/FR |
| | ethyl methyl-para-tolyl glycidate | FL/FR |
| (E)- | ethyl tiglate | FL/FR |
| | furfuryl propionate | FL |
| 2- | furyl pentyl ketone | FL |
| | geranyl butyrate | FL/FR |
| 2- | heptanol | FL/FR |
| 3- | heptyl dihydro-5-methyl-2(3H)-furanone | FL/FR |
| | hexanal propylene glycol acetal | FL/FR |
| 2- | hexen-1-ol | FL/FR |
| (Z)-3- | hexen-1-yl (E)-2-hexenoate | FL/FR |
| (E)-3- | hexen-1-yl acetate | FL/FR |
| (Z)-3- | hexen-1-yl isobutyrate | FL/FR |
| | hexyl acetate | FL/FR |
| | hexyl hexanoate | FL/FR |
| | kiwi distillates | FL |
| | lilac pentanol | FL/FR |
| 3- | mercapto-3-methyl-1-hexanol | |
| | methyl (E)-2-octenoate | FL/FR |
| | methyl (E)-3-nonenoate | FL |
| | methyl 3-hexenoate | FL/FR |
| | methyl 3-nonenoate | FL/FR |
| | methyl heptanoate | FL/FR |
| | methyl valerate | FL/FR |
| 3- | methyl-2-butenal | FL/FR |
| | neryl formate | FL/FR |
| (Z)-3- | nonen-1-yl acetate | FL |
| (Z)-5- | octen-1-yl acetate | FL |
| | octen-1-yl cyclopentanone | FL/FR |
| 2- | phenyl propionaldehyde dimethyl acetal | FL/FR |
| 1- | phenyl-2-pentanol | FL/FR |
| | pineapple pentenoate | FL/FR |
| iso | propyl 2-methyl butyrate | FL/FR |
| | propyl formate | FL/FR |
| | tetrahydrofurfuryl butyrate | FL/FR |
| | tiglaldehyde | FL/FR |
| green |
| | acetaldehyde dihexyl acetal | FL/FR |
| | actinidia chinensis fruit extract | FL/FR |
| | alfalfa distillates | FL |
| | allyl butyrate | FL/FR |
| iso | amyl isovalerate | FL/FR |
| iso | butyl isovalerate | FL/FR |
| | cuminyl acetaldehyde | FL/FR |
| | cyclohexyl formate | FL/FR |
| 3- | decen-2-one | FL/FR |
| 3,4- | dimethoxystyrene | FL |
| 2- | ethyl butyraldehyde | FL |
| | geranyl acetate | FL/FR |
| | geranyl isovalerate | FL/FR |
| | grape butyrate | FL/FR |
| | heptyl butyrate | FL/FR |
| | hexanol | FL/FR |
| (E)-3- | hexen-1-ol | FL/FR |
| (Z)-3- | hexen-1-yl (Z)-3-hexenoate | FL/FR |
| (Z)-3- | hexen-1-yl acetate | FL/FR |
| (E)-2- | hexen-1-yl acetate | FL/FR |
| (E)-2- | hexen-1-yl hexanoate | FL/FR |
| (Z)-3- | hexen-1-yl hexanoate | FL/FR |
| (Z)-3- | hexen-1-yl isovalerate | FL/FR |
| (Z)-3- | hexen-1-yl octanoate | FL/FR |
| (E)-2- | hexen-1-yl propionate | FL/FR |
| (Z)-3- | hexen-1-yl valerate | FL/FR |
| (E)-2- | hexen-1-yl valerate | FL/FR |
| 1- | hexen-3-ol | FL/FR |
| (E)-2- | hexenal diethyl acetal | FL |
| 3- | hexenyl 2-methyl butyrate | FL/FR |
| | hexyl (E)-tiglate | FL/FR |
| | hexyl benzoate | FL/FR |
| | hexyl butyrate | FL/FR |
| | hexyl isobutyrate | FL/FR |
| | hexyl isovalerate | FL/FR |
| | hexyl octanoate | FL/FR |
| iso | jasmone | FL/FR |
| iso | jasmone | FL/FR |
| | methyl (E)-3-hexenoate | FL/FR |
| | methyl 2-undecynoate | FL |
| | methyl octanoate | FL/FR |
| | methyl octine carbonate | FL/FR |
| | methyl R-3-acetoxyhexanoate | |
| 3-(5- | methyl-2-furyl) butanal | FL |
| 4- | methyl-2-pentenal | FL |
| | neryl butyrate | FL/FR |
| | neryl propionate | FL/FR |
| (E,E)-2,4- | octadienal | FL |
| 2,4- | octadienal | FL |
| 2- | octenyl acetate | FL/FR |
| | papaya isobutyrate | FL/FR |
| (E)-2- | pentenal | FL/FR |
| 2- | pentyl furan | FL/FR |
| | phenoxyethyl isobutyrate | FL/FR |
| 4-(1- | propenyl) pyridine | |
| | terpinyl propionate | FL/FR |
| | thiogeraniol | FL/FR |
| honey |
| | phenethyl acetate | FL/FR |
| nutty |
| | acetal | FL/FR |
| pungent |
| | acetaldehyde | FL |
| rummy |
| (E)-2- | hexen-1-yl formate | FL/FR |
| sour |
| 3- | methyl valeric acid | FL |
| sulfurous |
| | buchu mercaptan | FL/FR |
| tropical |
| beta- | cyclocitral | FL/FR |
| | guava distillates | FL |
| | propyl propionate | FL/FR |
| | psidium guajava fruit | FL |
| | psidium guajava fruit extract | FL/FR |
| vegetable |
| 2- | methyl valeraldehyde | FL/FR |
| waxy |
| iso | amyl butyrate | FL/FR |
| 9- | decenoic acid | FL/FR |
| 2- | nonanol | FL/FR |
| | octyl 2-methyl butyrate | FL/FR |
| | octyl butyrate | FL/FR |
| 3- | octyl formate | FL/FR |
| whiskey |
| 3- | methyl-1-pentanol | FL/FR |
| woody |
| beta- | damascenone | FL/FR |
| beta- | ionone | FL/FR |
| |
Potential Uses:
Occurrence (nature, food, other): noteSynonyms:
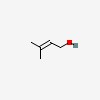
| | but-2-en-1-ol, 3-methyl- | | 2- | buten-1-ol, 3-methyl- | | | dimethyl allyl alcohol | | 3,3- | dimethylallyl alcohol | | 3- | methyl but-2-en-1-ol | | 3- | methyl crotyl alcohol | | 3- | methyl-2-buten-1-ol | | 2- | methyl-2-buten-4-ol | | 3- | methyl-2-butene-1-ol | | 3- | methyl-2-butenol | | 3- | methyl-2-butenyl alcohol | | 3- | methylbut-2-en-1-ol | | 3- | methylcrotyl alcohol | | | prenol (3-methyl-2-buten-1-ol) | | | prenyl alcohol | | | vertenol (Grau) |
Articles:
| PubMed: | MEP Pathway-mediated isopentenol production in metabolically engineered Escherichia coli. |
| PubMed: | Secondary ion mass spectrometry imaging of Dictyostelium discoideum aggregation streams. |
| PubMed: | cis-Prenyltransferase atCPT6 produces a family of very short-chain polyisoprenoids in planta. |
| PubMed: | Metabolic signatures of triatomine vectors of Trypanosoma cruzi unveiled by metabolomics. |
| PubMed: | Metabolic engineering of Escherichia coli for high-specificity production of isoprenol and prenol as next generation of biofuels. |
| PubMed: | Antibacterial/antifungal activity and synergistic interactions between polyprenols and other lipids isolated from Ginkgo biloba L. leaves. |
| PubMed: | N,O-Nucleosides from ene reactions of nitrosocarbonyl intermediates with the 3-methyl-2-buten-1-ol. |
| PubMed: | Sugar availability modulates polyisoprenoid and phytosterol profiles in Arabidopsis thaliana hairy root culture. |
| PubMed: | Synthesis and NMR characterization of (Z,Z,Z,Z,E,E,ω)-heptaprenol. |
| PubMed: | Polyprenols from the needles of Taxus chinensis var. mairei. |
| PubMed: | Bacterial degradation of tert-amyl alcohol proceeds via hemiterpene 2-methyl-3-buten-2-ol by employing the tertiary alcohol desaturase function of the Rieske nonheme mononuclear iron oxygenase MdpJ. |
| PubMed: | Hepatoprotective effects of polyprenols from Ginkgo biloba L. leaves on CCl4-induced hepatotoxicity in rats. |
| PubMed: | Role of novel terpenes in transcutaneous permeation of valsartan: effectiveness and mechanism of action. |
| PubMed: | Configuration of polyisoprenoids affects the permeability and thermotropic properties of phospholipid/polyisoprenoid model membranes. |
| PubMed: | Fragrance material review on 3-methyl-2-buten-1-ol. |
| PubMed: | Separation of hydrophobic metabolites using monolithic silica column in high-performance liquid chromatography and supercritical fluid chromatography. |
| PubMed: | Complete (1)H and (13)C signal assignment of prenol-10 with 3D NMR spectroscopy. |
| PubMed: | Solid-phase organic synthesis of polyisoprenoid alcohols with traceless sulfone linker. |
| PubMed: | A strategy for position-selective epoxidation of polyprenols. |
| PubMed: | Tumor-specificity and type of cell death induced by vitamin K2 derivatives and prenylalcohols. |
| PubMed: | Characterization of a PRL protein tyrosine phosphatase from Plasmodium falciparum. |
| PubMed: | The search for polyprenols in dendroflora of Vietnam. |
| PubMed: | Analysis of ubiquinones, dolichols, and dolichol diphosphate-oligosaccharides by liquid chromatography-electrospray ionization-mass spectrometry. |
| PubMed: | Antidyslipidemic activity of polyprenol from Coccinia grandis in high-fat diet-fed hamster model. |
| PubMed: | The use of long-chain plant polyprenols as a means to modify the biological properties of new biodegradable polyurethane scaffolds for tissue engineering. A pilot study. |
| PubMed: | Alloprenols: novel alpha-trans-polyprenols of Allophylus caudatus. |
| PubMed: | Precise bacterial polyprenol length control fails in Saccharomyces cerevisiae. |
| PubMed: | A new approach for the asymmetric total synthesis of umbelactone. |
| PubMed: | Activity of Pichia pastoris alternative cis-prenyltransferase is correlated with proliferation of peroxisomes. |
| PubMed: | Separation of polyprenol and dolichol by monolithic silica capillary column chromatography. |
| PubMed: | Polyprenyl lipid synthesis in mammalian cells expressing human cis-prenyl transferase. |
| PubMed: | The occurrence of polyprenols in seeds and leaves of woody plants. |
| PubMed: | Light conditions alter accumulation of long chain polyprenols in leaves of trees and shrubs throughout the vegetation season. |
| PubMed: | A comprehensive classification system for lipids. |
| PubMed: | Sulfone and phosphinic acid analogs of decaprenolphosphoarabinose as potential anti-tuberculosis agents. |
| PubMed: | [Studies on purification of polyprenol from Ginkgo biloba L. leaves]. |
| PubMed: | Analysis of the volatile aroma constituents of parental and hybrid clones of pepino (Solanum muricatum). |
| PubMed: | Proteins are polyisoprenylated in Arabidopsis thaliana. |
| PubMed: | Defects in the N-linked oligosaccharide biosynthetic pathway in a Trypanosoma brucei glycosylation mutant. |
| PubMed: | Heteromeric geranyl diphosphate synthase from mint: construction of a functional fusion protein and inhibition by bisphosphonate substrate analogs. |
| PubMed: | Photooxygenation of allylic alcohols: kinetic comparison of unfunctionalized alkenes with prenol-type allylic alcohols, ethers and acetates. |
| PubMed: | Chemical analysis of Ginkgo biloba leaves and extracts. |
| PubMed: | Protein farnesyltransferase and protein prenylation in Plasmodium falciparum. |
| PubMed: | A general stereocontrolled, convergent synthesis of oligoprenols that parallels the biosynthetic pathway. |
| PubMed: | Biosynthesis of a substituted cellulose from a mutant strain of Xanthomonas campestris. |
| PubMed: | [Synthesis of dolicyl- and polyprenyl sulfates]. |
| PubMed: | [Mechanisms of action of aerosol preparations based on Abies siberica polyprenols in experimental influenza infection]. |
| PubMed: | [Prophylactic efficacy of aerosol preparations based on Abies siberica polyprenols in experimental influenza infection]. |
| PubMed: | Constituents of Pilocarpus trachylophus. |
| PubMed: | Rational design of enantioselective enzymes requires considerations of entropy. |
| PubMed: | High-resolution analysis of polyprenols by supercritical fluid chromatography. |
| PubMed: | Study on the biosynthesis of dolichol in yeast: recognition of the prenyl chain length in polyprenol reduction. |
| PubMed: | Polyprenols in hairy roots of Coluria geoides. |
| PubMed: | Incorporation of radiolabeled prenyl alcohols and their analogs into mammalian cell proteins. A useful tool for studying protein prenylation. |
| PubMed: | The diversity of polyprenol pattern in leaves of fruit trees belonging to Rosaceae and Cornaceae. |
| PubMed: | Nonglucosylated oligosaccharides are transferred to protein in MI8-5 Chinese hamster ovary cells. |
| PubMed: | Transfer of two oligosaccharides to protein in a Chinese hamster ovary cell B211 which utilizes polyprenol for its N-linked glycosylation intermediates. |
| PubMed: | Identification of Schizosaccharomyces pombe prenol as dolichol-16,17. |
| PubMed: | Reduced utilization of Man5GlcNAc2-P-P-lipid in a Lec9 mutant of Chinese hamster ovary cells: analysis of the steps in oligosaccharide-lipid assembly. |
| PubMed: | Prenylation of proteins in Trypanosoma brucei. |
| PubMed: | On the specific pattern of long chain polyprenols in green needles of Pinus mugo Turra. |
| PubMed: | 6-O-alpha-L-Arabinopyranosyl-beta-D-glucopyranosides as aroma precursors from passion fruit. |
| PubMed: | Dolichyl phosphate, a potent inducer of apoptosis in rat glioma C6 cells. |
| PubMed: | Biosynthesis of isoprenoid compounds in cattle filarial parasite Setaria digitata. |
| PubMed: | Biosynthesis of membrane-derived oligosaccharides. Membrane-bound glucosyltransferase system from Escherichia coli requires polyprenyl phosphate. |
| PubMed: | Function of alpha-D-glucosyl monophosphorylpolyprenol in biosynthesis of cell wall teichoic acids in Bacillus coagulans. |
| PubMed: | The function of galactosyl phosphorylpolyprenol in biosynthesis of lipoteichoic acid in Bacillus coagulans. |
| PubMed: | Long-chain polyprenols in gymnosperm plants. |
| PubMed: | Prenol nomenclature. Recommendations 1986. IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN). |
| PubMed: | Biosynthesis of poly(galactosylglycerol phosphate) in Bacillus coagulans. |
| PubMed: | Structure and functions of linkage unit intermediates in the biosynthesis of ribitol teichoic acids in Staphylococcus aureus H and Bacillus subtilis W23. |
| PubMed: | Total synthesis of chain-length-uniform dolichyl phosphates and their fitness to accept hexoses in the enzymatic formation of lipoglycans. |
| PubMed: | Biosynthesis of wall teichoic acids in Staphylococcus aureus H, Micrococcus varians and Bacillus subtilis W23. Involvement of lipid intermediates containing the disaccharide N-acetylmannosaminyl N-acetylglucosamine. |
| PubMed: | The occurrence and seasonal distribution of C50-C60-polyprenols and of C100-and similar long-chain polyprenols in leaves of plants. |
| PubMed: | Subcellular localization and substrate specificity of dolichol kinase from rat liver. |
| PubMed: | Biosynthesis of polysaccharides in Acetobacter xylinum. Sequential synthesis of a heptasaccharide diphosphate prenol. |
| PubMed: | Synthesis of mannosyl cellobiose diphosphate prenol in Acetobacter xylinum. |
| PubMed: | Paramagnetic isoprenoid carrier lipids. 1. Chemical synthesis and incorporation into model membranes. |
| PubMed: | Factors affecting glucosyl and mannosyl transfer to dolichyl monophosphate by liver cell-free preparations. |
| PubMed: | Glucolipids of Zea mays and Pisum sativum. |
| PubMed: | Mechanism of the isomerization of isopentenyl pyrophosphate in Rhodotorual rubra-1. |
| PubMed: | Incorporation of radioactive mevalonate into C50 and C55 phenols by Streptococcus mutans. |
| PubMed: | Identification of prenol intermediates of wall biosynthesis in growing cells of Lactobacillus plantarum. |
| PubMed: | The transfer of mannose from guanosine diphosphate mannose to dolichol phosphate and protein by pig liver endoplasmic reticulum. |
| PubMed: | Polyprenol phosphate as an acceptor of mannose from guanosine diphosphate mannose in Aspergillus niger. |
| PubMed: | Polyprenols of Aspergillus niger. Their characterization, biosynthesis and subcellular distribution. |
| PubMed: | Prenol phosphates and mannosyltransferases. |
| PubMed: | The characterization of undecaprenol of Lactobacillus plantarum. |
| PubMed: | The characterization and distribution of hexahydropolyprenyl esters in cultures of Aspergillus fumigatus Fresenius. |
| PubMed: | Testicular sterols. V. Preparation and partial purification of a microsomal prenol pyrophosphate pyrophosphohydrolase. |
|
 3D/inchi
3D/inchi













