Articles:
(S)-p-mentha-1,8-dien-7-al
Notes:
None found
| CAS Number: | 18031-40-8 | 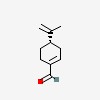 3D/inchi 3D/inchi
|
| FDA UNII: | 5EL0Y7P6LP | |
| Nikkaji Web: | J132.738K | |
| Beilstein Number: | 2326448 | |
| MDL: | MFCD00001543 | |
| XlogP3-AA: | 2.60 (est) | |
| Molecular Weight: | 150.22078000 | |
| Formula: | C10 H14 O | |
| NMR Predictor: | Predict (works with chrome or firefox) | |
Category: flavor and fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
| Google Scholar: | Search |
| Google Books: | Search |
| Google Scholar: with word "volatile" | Search |
| Google Scholar: with word "flavor" | Search |
| Google Scholar: with word "odor" | Search |
| Perfumer and Flavorist: | Search |
| Google Patents: | Search |
| US Patents: | Search |
| EU Patents: | Search |
| Pubchem Patents: | Search |
| PubMed: | Search |
| NCBI: | Search |
| FEMA Number: | 3557 (S)-p-mentha-1,8-dien-7-al |
| FDA: | No longer provide for the use of these seven synthetic flavoring substances |
Physical Properties:
| Appearance: | pale yellow to dark yellow clear oily liquid (est) |
| Assay: | 92.00 to 100.00 % |
| Food Chemicals Codex Listed: | No |
| Specific Gravity: | 0.95800 to 0.97300 @ 25.00 °C. |
| Pounds per Gallon - (est).: | 7.972 to 8.096 |
| Refractive Index: | 1.50700 to 1.51500 @ 20.00 °C. |
| Optical Rotation: | -118.00 to -125.00 |
| Boiling Point: | 104.00 to 105.00 °C. @ 10.00 mm Hg |
| Boiling Point: | 237.00 °C. @ 760.00 mm Hg |
| Vapor Pressure: | 0.043000 mmHg @ 25.00 °C. (est) |
| Flash Point: | 203.00 °F. TCC ( 95.00 °C. ) |
| logP (o/w): | 3.130 |
| Soluble in: | |
| alcohol | |
| water, 160.7 mg/L @ 25 °C (est) | |
| Insoluble in: | |
| water | |
Organoleptic Properties:
| Odor Type: herbal | |
| Odor Strength: | medium , recommend smelling in a 10.00 % solution or less |
| Substantivity: | 8 hour(s) at 100.00 % |
| fresh green oily grassy fatty minty cherry | |
| Odor Description: at 10.00 % in dipropylene glycol. | fresh green oily grassy fatty mint cherry |
| Flavor Type: aromatic | |
| aromatic sweet woody spicy waxy orange lime aldehydic | |
| Taste Description: | aromatic sweet woody spicy waxy orange lime aldehydic |
| Odor and/or flavor descriptions from others (if found). | |
Cosmetic Information:
| None found |
Suppliers:
| Alfa Biotechnology |
| For experimental / research use only. |
| (-)-Perillaldehyde 98% |
| BOC Sciences |
| For experimental / research use only. |
| Perillaldehyde >98% |
| Fuzhou Farwell |
| (-)-Perillaldehyde |
| Santa Cruz Biotechnology |
| For experimental / research use only. |
| (S)-(-)-Perillaldehyde |
| Sigma-Aldrich |
| (S)-(-)-Perillaldehyde, ≥92%
Odor: cherry; oily; green |
| Certified Food Grade Products |
| TCI AMERICA |
| For experimental / research use only. |
| (-)-Perillaldehyde >90.0%(GC) |
Safety Information:
| Preferred SDS: View | |
| European information : | |
| Most important hazard(s): | |
| Xi - Irritant | |
|
R 36/37/38 - Irritating to eyes, respiratory system, and skin. S 02 - Keep out of the reach of children. S 24/25 - Avoid contact with skin and eyes. S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S 37/39 - Wear suitable gloves and eye/face protection. | |
| Hazards identification | |
| Classification of the substance or mixture | |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) | |
| None found. | |
| GHS Label elements, including precautionary statements | |
| Pictogram | |
| Hazard statement(s) | |
| None found. | |
| Precautionary statement(s) | |
| None found. | |
| Human Experience: | |
| 1 % solution: no irritation or sensitization. | |
| Oral/Parenteral Toxicity: | |
| Not determined | |
| Dermal Toxicity: | |
| Not determined | |
| Inhalation Toxicity: | |
| Not determined | |
Safety in Use Information:
| Category: | flavor and fragrance agents | ||
| RIFM Fragrance Material Safety Assessment: Search | |||
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice | |||
| IFRA Critical Effect: | Dermal sensitization | ||
| IFRA: | View Standard | ||
| View IFRA Standards Library for complete information. | |||
| Please review Amendment 49 IFRA documentation for complete information. | |||
| IFRA RESTRICTION LIMITS IN THE FINISHED PRODUCT (%): | |||
| Category 1: Products applied to the lips | |||
| 0.054 % | |||
| Category 2: Products applied to the axillae | |||
| 0.016 % | |||
| Category 3: Products applied to the face/body using fingertips | |||
| 0.32 % | |||
| Category 4: Products related to fine fragrance | |||
| 0.30 % | |||
| Category 5: Products applied to the face and body using the hands (palms), primarily leave-on | |||
| Category 5A: Body lotion products applied to the body using the hands (palms), primarily leave-on | |||
| 0.076 % | |||
| Category 5B: Face moisturizer products applied to the face using the hands (palms), primarily leave-on | |||
| 0.076 % | |||
| Category 5C: Hand cream products applied to the hands using the hands (palms), primarily leave-on | |||
| 0.076 % | |||
| Category 5D: Baby Creams, baby Oils and baby talc | |||
| 0.076 % | |||
| Category 6: Products with oral and lip exposure | |||
| 0.18 % | |||
| Category 7: Products applied to the hair with some hand contact | |||
| Category 7A: Rinse-off products applied to the hair with some hand contact | |||
| 0.61 % | |||
| Category 7B: Leave-on products applied to the hair with some hand contact | |||
| 0.61 % | |||
| Category 8: Products with significant anogenital exposure | |||
| 0.032 % | |||
| Category 9: Products with body and hand exposure, primarily rinse off | |||
| 0.59 % | |||
| Category 10: Household care products with mostly hand contact | |||
| Category 10A: Household care excluding aerosol products (excluding aerosol/spray products) | |||
| 2.10 % | |||
| Category 10B: Household aerosol/spray products | |||
| 2.10 % | |||
| Category 11: Products with intended skin contact but minimal transfer of fragrance to skin from inert substrate | |||
| Category 11A: Products with intended skin contact but minimal transfer of fragrance to skin from inert substrate without UV exposure | |||
| 1.20 % | |||
| Category 11B: Products with intended skin contact but minimal transfer of fragrance to skin from inert substrate with potential UV exposure | |||
| 1.20 % | |||
| Category 12: Products not intended for direct skin contact, minimal or insignificant transfer to skin | |||
| No Restriction | |||
| Notes: | |||
| IFRA FLAVOR REQUIREMENTS: | |||
Due to the possible ingestion of small amounts of fragrance ingredients from their use in products in Categories 1 and 6, materials must not only comply with IFRA Standards but must also be recognized as safe as a flavoring ingredient as defined by the IOFI Code of Practice (www.iofi.org). For more details see chapter 1 of the Guidance for the use of IFRA Standards. | |||
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). | |||
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library | |||
| publication number: 11 | |||
| Click here to view publication 11 | |||
| average usual ppm | average maximum ppm | ||
| baked goods: | - | 4.50000 | |
| beverages(nonalcoholic): | - | 4.00000 | |
| beverages(alcoholic): | - | 6.00000 | |
| breakfast cereal: | - | - | |
| cheese: | - | - | |
| chewing gum: | - | - | |
| condiments / relishes: | - | - | |
| confectionery froastings: | - | - | |
| egg products: | - | - | |
| fats / oils: | - | - | |
| fish products: | - | - | |
| frozen dairy: | - | 6.30000 | |
| fruit ices: | - | - | |
| gelatins / puddings: | - | 4.50000 | |
| granulated sugar: | - | - | |
| gravies: | - | - | |
| hard candy: | - | - | |
| imitation dairy: | - | - | |
| instant coffee / tea: | - | - | |
| jams / jellies: | - | - | |
| meat products: | - | 20.00000 | |
| milk products: | - | - | |
| nut products: | - | - | |
| other grains: | - | - | |
| poultry: | - | - | |
| processed fruits: | - | - | |
| processed vegetables: | - | - | |
| reconstituted vegetables: | - | - | |
| seasonings / flavors: | - | - | |
| snack foods: | - | - | |
| soft candy: | - | 9.70000 | |
| soups: | - | - | |
| sugar substitutes: | - | - | |
| sweet sauces: | - | - | |
Safety References:
| Flavor & Extract Manufacturers Association (FEMA) reference(s): | |
| The FEMA GRAS assessment of alicyclic substances used as flavor ingredients. View pdf | |
| EPI System: | View |
| Chemical Carcinogenesis Research Information System: | Search |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 2724159 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WGK Germany: | 3 |
| (4S)-4-prop-1-en-2-ylcyclohexene-1-carbaldehyde | |
| Chemidplus: | 0018031408 |
| RTECS: | GW2967200 for cas# 18031-40-8 |
References:
| (4S)-4-prop-1-en-2-ylcyclohexene-1-carbaldehyde | |
| NIST Chemistry WebBook: | Search Inchi |
| Pubchem (cid): | 2724159 |
| Pubchem (sid): | 134992495 |
| Flavornet: | 18031-40-8 |
| Pherobase: | View |
Other Information:
| (IUPAC): | Atomic Weights of the Elements 2011 (pdf) |
| Videos: | The Periodic Table of Videos |
| tgsc: | Atomic Weights use for this web site |
| (IUPAC): | Periodic Table of the Elements |
| CHEMBL: | View |
| Metabolomics Database: | Search |
| HMDB (The Human Metabolome Database): | Search |
| Export Tariff Code: | 2912.29.6000 |
| VCF-Online: | VCF Volatile Compounds in Food |
| ChemSpider: | View |
| Wikipedia: | View |
Potential Blenders and core components note
Potential Uses:
| apple | FR | |
| citrus | FR | |
| cumin oil replacer | FR | |
| grapefruit | FR | |
| grass | ||
| green | FR | |
| herbal | FR | |
| lime | FR | |
| mint | FR | |
| orange | FR | |
| spearmint | FR | |
| tea green tea | FR | |
| woody | FR |
Occurrence (nature, food, other): note
| blackberry fruit Search PMC Picture | |
| grapefruit juice Search Trop Picture | |
| grapefruit oil Search Trop Picture | |
| labdanum siam Search PMC Picture | |
| lime fruit juice Search Trop Picture | |
| lime peel oil Search Trop Picture | |
| mandarin peel Search Trop Picture | |
| orange oil Search Trop Picture | |
| peppermint Search Trop Picture | |
| tea black tea Search Trop Picture |
Synonyms:
| 1- | cyclohexene-1-carboxaldehyde, 4-(1-methylethenyl)-, (4S)- |
| 1- | cyclohexene-1-carboxaldehyde, 4-(1-methylethenyl)-, (S)- |
| 1- | cyclohexene-1-carboxaldehyde, 4-isopropenyl-, (S)-(-)- |
| (S)-(-)- | dihydrocuminyl aldehyde |
| (S)-p- | mentha-1,8-dien-7-al |
| (S)-para- | mentha-1,8-dien-7-al |
| (S)-(-)4-(1- | methyl ethenyl)-1-cyclohexene-1-carboxaldehyde |
| (S)-(-)4-(1- | methylethenyl)-1-cyclohexene-1-carboxaldehyde |
| (4S)-4-(1- | methylvinyl)cyclohex-1-enecarbaldehyde |
| (-)- | perilla aldehyde |
| (S)- | perilla aldehyde |
| (S)-(-)- | perilla aldehyde |
| (-)- | perillaaldehyde |
| (S)- | perillaaldehyde |
| (S)-(-)- | perillaaldehyde |
| (-)- | perillaldehyde |
| (S)-(-)- | perillaldehyde |
| L- | perillaldehyde |
| (4S)-4-( | prop-1-en-2-yl)cyclohex-1-ene-1-carbaldehyde |
| (S)-4-( | prop-1-en-2-yl)cyclohex-1-enecarbaldehyde |
| (4S)-4- | prop-1-en-2-ylcyclohexene-1-carbaldehyde |
| (4S)-4-iso | propenyl-1-cyclohexene-1-carbaldehyde |
| (S)-(-)-4-iso | propenyl-1-cyclohexene-1-carboxaldehyde |
| L-4-iso | propenyl-1-cyclohexene-1-carboxaldehyde |
| laevo-4-iso | propenyl-1-cyclohexene-1-carboxaldehyde |
| (S)-4-iso | propenyl-cyclohex-1-enecarbaldehyde |
| (S)-4-iso | propenyl-cyclohexene-1-carboxaldehyde |
| (4S)-4-iso | propenylcyclohex-1-ene-1-carbaldehyde |


