|
Category: flavor and fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
Physical Properties:
| Appearance: | yellow clear liquid (est) |
| Assay: | 98.00 to 100.00 %
|
| Food Chemicals Codex Listed: | No |
| Specific Gravity: | 0.94600 to 0.95200 @ 25.00 °C.
|
| Pounds per Gallon - (est).: | 7.872 to 7.922
|
| Refractive Index: | 1.51000 to 1.51400 @ 20.00 °C.
|
| Boiling Point: | 274.00 to 275.00 °C. @ 760.00 mm Hg
|
| Acid Value: | 1.00 max. KOH/g
|
| Vapor Pressure: | 0.020000 mmHg @ 20.00 °C. |
| Flash Point: | > 212.00 °F. TCC ( > 100.00 °C. )
|
| logP (o/w): | 4.042 (est) |
| Soluble in: |
| | alcohol | | | water, 12.48 mg/L @ 25 °C (est) |
| Stability: |
| | antiperspirant | | | cream | | | deodorant spray | | | detergent: traces - 0.2% | | | hair spray | | | shampoo: traces - 0.1% | | | soap: traces - 0.1% | | | APC: traces - 0.2% | | | candle: traces - 0.2% | | | fine fragrances: traces - 0.1% | | | shower gel: traces - 0.1% | | | softener: traces - 0.2% |
Organoleptic Properties:
| |
| Odor Type: floral |
| |
| Odor Strength: | high ,
recommend smelling in a 10.00 % solution or less |
| |
| Substantivity: | 216 hour(s) at 100.00 % |
| |
| | natural sweet fruity rose plum grape raspberry sugar |
Odor Description:
at 10.00 % in dipropylene glycol. | natural sweet fruity rose plum grape raspberry sugar
Luebke, William tgsc, (1987) |
| |
| | woody sweet fruity earthy green floral |
Odor Description:
| Woody, sweet, fruity, earthy with green floral nuances
Mosciano, Gerard P&F 16, No. 1, 31, (1991) |
| |
| |
| Flavor Type: woody |
| |
| | woody floral herbal green fruity spicy tobacco |
Taste Description:
at 20.00 ppm. | Woody, floral, herbal, green and fruity with spicy tobacco nuances
Mosciano, Gerard P&F 16, No. 1, 31, (1991) |
| |
| Odor and/or flavor descriptions from others (if found). |
| |
| Firmenich |
| DAMASCENONE for fragrance |
| Odor Description: | A very powerful floral fruity note. DAMASCENONE smells intensely natural, rose, plum, berry and tobacco
DAMASCENONE gives attractive and beautiful nuances to all fragrance types even at trace or low levels. |
| |
| Firmenich |
| DAMASCENONE TOTAL for fragrance |
| Odor Description: | A very powerful floral fruity note that is very close in quality to DAMASCENONE. DAMASCENONE TOTAL smells intensely natural, rose, plum, berry and tobacco
DAMASCENONE TOTAL gives attractive and beautiful nuances to all fragrance types even at trace or low levels and can often be used as a direct replacement of DAMASCENONE. |
| |
| Sigma-Aldrich |
| Damascenone, natural, 1.1-1.3�wt. % (190 proof ethanol), FG |
| Odor Description: | apple; woody; herbaceous; nutty; citrus; rose; smoky; wine-like. |
| |
| Moellhausen |
| DAMASCENONE 93% |
| Odor Description: | persistent fruity, floral; strawberry- and rose-notes, tobacco. |
| Taste Description: | woody, floral, herbal, green and fruity with spicy tobacco notes. |
| |
| Alfrebro |
| Damascenone 1% in ETOH, Natural |
| Odor Description: | Natural Sweet Fruity Rose Plum Grape Raspberry Sugar |
| |
| IFF |
| Damascenone 93% Min. |
| Odor Description: | Floral rosy, fruity odor with plum, berry and tobacco nuances |
| |
| IFF |
| Damascenone 98% Min. |
| Odor Description: | Floral rosy, fruity odor with plum, berry and tobacco nuances |
| |
| Ernesto Ventós |
| DAMASCENONA IFF 95% |
| Odor Description: | Floral rosy, fruity odor with plum, berry and tobacco nuances |
| |
| Firmenich |
| DAMASCENONE Kosher for flavor |
| Odor Description: | A very powerful floral fruity note. DAMASCENONE smells intensely natural, rose, plum, berry and tobacco |
| Taste Description: | Fruity floral quince rosy and cooked notes
DAMASCENONE is an essential heart note for fruit with cooked and jammy nuances.
It also performs well in wine, tobacco and tea flavors |
| |
| |
Cosmetic Information:
Suppliers:
| ACS International |
| Rosenone Total
Odor: floral woody sweet rose plum grape |
| Operational Capabilities |
| ACS International |
| Rosenone
Odor: floral woody sweet rose plum grape |
| Advanced Biotech |
| BETA DAMASCENONE 1% ETOH NATURAL
|
| Advanced Biotech |
| BETA DAMASCENONE 1% TAC NAT
|
| Advanced Biotech |
| BETA DAMASCENONE 5% PG NATURAL
|
| Advanced Biotech |
| BETA DAMASECNONE 1% ETOH (S TYPE)
|
| Advanced Biotech |
| FERMENTONE 1% IN PG NATURAL
|
| Advanced Biotech |
| FERMENTONE 5% ETOH NATURAL
|
| Advanced Biotech |
| FERMENTONE 5% ETOH NATURAL
|
| Advanced Biotech |
| FERMENTONE 5% PG NATURAL
|
| Advanced Biotech |
| FERMENTONE CONCENTRATE NATURAL
|
| Advanced Biotech |
| FERMENTONE EXTRA 1% IN TRIACETIN NATURAL
Odor: Berry |
| Advanced Biotech |
| FERMENTONE EXTRA 1% PG NATURAL
|
| Advanced Biotech |
| FERMENTONE EXTRA 5% TRIACETIN NATURAL
|
| Advanced Biotech |
| FERMENTONE EXTRA NATURAL
Odor: Berry |
| Advanced Biotech |
| FERMENTONE NATURAL
1.25% min. Odor: Berry |
| Advanced Biotech |
| FERMENTONE SUPRA 1% IN TRIACETIN NATURAL
|
| Advanced Biotech |
| ROASTAROME NATURAL
|
| Advanced Biotech |
| SYNTHETIC BETA DAMASCENONE
|
| Alfrebro |
| Damascenone 1% in ETOH, Natural
Odor: Natural Sweet Fruity Rose Plum Grape Raspberry Sugar |
| Alfrebro |
| Damascenone 1% in PG, Natural
|
| Associate Allied Chemicals |
| Damascenone
|
| About |
| Augustus Oils |
| Damascenone
|
| Services |
| Axxence Aromatic |
| beta-DAMASCENONE 1,5% IN ETHANOL, Natural
Kosher |
| Sustainability |
| Axxence Aromatic |
| beta-DAMASCENONE, NATURAL Kosher
|
| Axxence Aromatic |
| DAMASCENONE 1% IN ET, Natural
Kosher |
| Axxence Aromatic |
| DAMASCENONE 1% IN PG, Natural
Kosher |
| Azelis UK |
| DAMASCENONE TOTAL
|
| Azelis UK |
| DAMASCENONE
|
| Berjé |
| Dascenone
|
| Media |
| BOC Sciences |
| For experimental / research use only. |
| Damascenone >98%
|
| De Monchy Aromatics |
| Damascenone
|
| Diffusions Aromatiques |
| DAMASCENONE
|
| Ernesto Ventós |
| DAMASCENONA IFF 95%
|
| Ernesto Ventós |
| DAMASCENONA IFF TOCO 98%
|
| Ernesto Ventós |
| DAMASCENONA IFF TOTAL TOCO 93%
|
| Ernesto Ventós |
| DAMASCENONE FIRMENICH 937450
Odor: POWERFUL, FLORAL, FRUITY |
| Ernesto Ventós |
| DAMASCENONE TOTAL FIRMENICH 937459
Odor: POWERFUL, FLORAL, FRUITY |
| Excellentia International |
| beta-Damascenone Natural
|
| FCI SAS |
| DAMASCENE
Odor: Floral, fruity, rose |
| Firmenich |
| DAMASCENONE FAB Kosher
for flavor Flavor: Fruity woody floral quince cooked fruit and tobacco notes DAMASCENONE FAB performs well in all kind of fruits including tomato, alcoholic notes, tea and floral flavors |
| Firmenich |
| DAMASCENONE Kosher
for flavor Flavor: Fruity floral quince rosy and cooked notes DAMASCENONE is an essential heart note for fruit with cooked and jammy nuances.
It also performs well in wine, tobacco and tea flavors |
| Firmenich |
| DAMASCENONE TOTAL
for fragrance Odor: A very powerful floral fruity note that is very close in quality to DAMASCENONE. DAMASCENONE TOTAL smells intensely natural, rose, plum, berry and tobacco Use: DAMASCENONE TOTAL gives attractive and beautiful nuances to all fragrance types even at trace or low levels and can often be used as a direct replacement of DAMASCENONE. |
| Firmenich |
| DAMASCENONE
for fragrance Odor: A very powerful floral fruity note. DAMASCENONE smells intensely natural, rose, plum, berry and tobacco Use: DAMASCENONE gives attractive and beautiful nuances to all fragrance types even at trace or low levels. |
| IFF |
| Damascenone 93% Min.
Odor: Floral rosy, fruity odor with plum, berry and tobacco nuances |
| IFF |
| Damascenone 98% Min.
Odor: Floral rosy, fruity odor with plum, berry and tobacco nuances |
| Indukern F&F |
| DAMASCENONE PURE
Odor: FLORAL, FRUITY, ROSE |
| Indukern F&F |
| DAMASCENONE TOTAL
Odor: FLORAL, FRUITY, ROSE |
| Liaison Carbone |
| DAMASCENONE 937450
|
| Lluch Essence |
| BETA-DAMASCENONE 95%
|
| Lluch Essence |
| BETA-DAMASCENONE 96%
Odor: WOODY, FLORAL, HERBACEOUS |
| Lluch Essence |
| BETA-DAMASCENONE NATURAL 1% ETHANOL
|
| Lluch Essence |
| BETA-DAMASCENONE NATURAL 1% PG
|
| M&U International |
| DAMASCENONE TOTAL
|
| M&U International |
| Damascenone
|
| Moellhausen |
| DAMASCENONE 10%DPG
|
| Moellhausen |
| DAMASCENONE 93%
Odor: persistent fruity, floral; strawberry- and rose-notes, tobacco. Flavor: woody, floral, herbal, green and fruity with spicy tobacco notes. |
| Moellhausen |
| DAMASCENONE 98%
|
| Oamic Ingredients |
| Beta Damascenone
Odor: Floral, fruity |
| Omega Ingredients |
| Damascenone 1.5% in Alcohol
|
| OQEMA |
| Damascenone
|
| OQEMA |
| Damascone Total
|
| Organica Aromatics |
| DAMASCENONE
Min 95% Odor: Fruity rose like character Use: It is very valuable constituent in floral, fruity aroma fine fragrances. |
| PCW France |
| Damascenone Total
|
| Steps to a fragranced product |
| PCW France |
| Damascenone
|
| Penta International |
| DAMASCENONE NATURAL 1% IN ETHANOL
|
| Penta International |
| DAMASCENONE NATURAL 1% IN PROPYLENE GLYCOL
|
| Penta International |
| DAMASCENONE NATURAL 1% IN TRIACETIN
|
| Penta International |
| DAMASCENONE NATURAL NEAT (Any Dilution in any solvent is available. Contact your customer service representative.)
|
| Penta International |
| DAMASCENONE TOTAL
|
| Penta International |
| DAMASCENONE
|
| Phoenix Aromas & Essential Oils |
| Beta-Damascenone Natural
|
| Phoenix Aromas & Essential Oils |
| Beta-Damascenone
|
| Prodasynth |
| DAMASCENONE
(> 98%) |
| R C Treatt & Co Ltd |
| Damascenone
|
| Reincke & Fichtner |
| Damascenone
|
| Riverside Aromatics |
| DAMASCENONE
|
| Riverside Aromatics |
| NATURAL DAMASCENONE FRACTION, 1% IN ETHANOL
|
| Riverside Aromatics |
| NATURAL DAMASCENONE FRACTION, 1% IN PG
|
| Riverside Aromatics |
| NATURAL DAMASCENONE FRACTION, 1% IN TRIACETIN
|
| Robertet |
| DAMASCENONE BETA AT 1% IN ETHANOL
Pure & Nat (EU) |
| Seasons and Harvest / Crop calendar |
| Robertet |
| DAMASCENONE BETA AT 1% IN PG
Pure & Nat (EU) |
| Robertet |
| DAMASCENONE BETA KEY 100
Pure & Nat (EU) |
| Robertet |
| DAMASCENONE BETA KEY 500 HALAL IN PG
Pure & Nat (EU) |
| Robertet |
| DAMASCENONE BETA KEY 500
Pure & Nat (EU) |
| Sigma-Aldrich |
| Damascenone, natural, 1.1-1.3�wt. % (190 proof ethanol), FG
Odor: apple; woody; herbaceous; nutty; citrus; rose; smoky; wine-like. |
| Certified Food Grade Products |
| SRS Aromatics |
| DAMASCENONE
|
| Stort Chemicals |
| Rosenone
|
| Synerzine |
| BETA-DAMASCENONE, NATURAL (5% IN PROPYLENE GLYCOL)
|
| Taytonn ASCC |
| Damascenone
Odor: Floral, Fruity, Styrax |
| Vigon International |
| DAMASCENONE BETA NATURAL 1% IN ETOH
|
| Vigon International |
| DAMASCENONE BETA NATURAL 5% IN ETOH
|
| Vigon International |
| DAMASCENONE BETA NATURAL 5% IN PG
|
| Vigon International |
| DAMASCENONE BETA NATURAL 5% IN TRIACETIN
|
| Vigon International |
| Damascenone Total
Odor: A very powerful floral fruity note that is very close in quality to DAMASCENONE. DAMASCENONE TOTAL smells intensely natural, rose, plum, berry and tobacco |
| Vigon International |
| Damascenone
Odor: FRUITY, FLORAL |
Safety Information:
| Preferred SDS: View |
| European information : |
| Most important hazard(s): | | Xi - Irritant |
R 36/37/38 - Irritating to eyes, respiratory system, and skin.
S 02 - Keep out of the reach of children.
S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S 36 - Wear suitable protective clothing.
|
| |
| Hazards identification |
| |
| Classification of the substance or mixture |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) |
| None found. |
| GHS Label elements, including precautionary statements |
| |
| Pictogram | |
| |
| Hazard statement(s) |
| None found. |
| Precautionary statement(s) |
| None found. |
| Oral/Parenteral Toxicity: |
|
Not determined
|
| Dermal Toxicity: |
|
Not determined
|
| Inhalation Toxicity: |
|
Not determined
|
Safety in Use Information:
| Category: | flavor and fragrance agents |
| RIFM Fragrance Material Safety Assessment: Search |
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice |
| IFRA Critical Effect: | Dermal sensitization |
| IFRA fragrance material specification: | | | Should not be used as fragrance ingredients such that the total level in
finished cosmetic products exceeds 0.02%, individually or in
combination. Based on test data showing sensitising potential for these
materials and on evidence of cross-reactivity. (IFRA guidelines) |
| IFRA: | View Standard |
| View IFRA Standards Library for complete information. |
| Please review Amendment 49 IFRA documentation for complete information. |
| IFRA RESTRICTION LIMITS IN THE FINISHED PRODUCT (%): |
| Category 1: Products applied to the lips |
| 0.0077 % |
| Category 2: Products applied to the axillae |
| 0.0023 % |
| Category 3: Products applied to the face/body using fingertips |
| 0.046 % |
| Category 4: Products related to fine fragrance |
| 0.043 % |
| | Category 5: Products applied to the face and body using the hands (palms), primarily leave-on |
| Category 5A: Body lotion products applied to the body using the hands (palms), primarily leave-on |
| 0.011 % |
| Category 5B: Face moisturizer products applied to the face using the hands (palms), primarily leave-on |
| 0.011 % |
| Category 5C: Hand cream products applied to the hands using the hands (palms), primarily leave-on |
| 0.011 % |
| Category 5D: Baby Creams, baby Oils and baby talc |
| 0.011 % |
| Category 6: Products with oral and lip exposure |
| 0.025 % |
| | Category 7: Products applied to the hair with some hand contact |
| Category 7A: Rinse-off products applied to the hair with some hand contact |
| 0.088 % |
| Category 7B: Leave-on products applied to the hair with some hand contact |
| 0.088 % |
| Category 8: Products with significant anogenital exposure |
| 0.0045 % |
| Category 9: Products with body and hand exposure, primarily rinse off |
| 0.084 % |
| | Category 10: Household care products with mostly hand contact |
| Category 10A: Household care excluding aerosol products (excluding aerosol/spray products) |
| 0.30 % |
| Category 10B: Household aerosol/spray products |
| 0.30 % |
| | Category 11: Products with intended skin contact but minimal transfer of fragrance to skin from inert substrate |
| Category 11A: Products with intended skin contact but minimal transfer of fragrance to skin from inert substrate without UV exposure |
| 0.17 % |
| Category 11B: Products with intended skin contact but minimal transfer of fragrance to skin from inert substrate with potential UV exposure |
| 0.17 % |
| Category 12: Products not intended for direct skin contact, minimal or insignificant transfer to skin |
| No Restriction |
| | Notes: |
| IFRA FLAVOR REQUIREMENTS: |
Due to the possible ingestion of small amounts of fragrance ingredients from their use in products in Categories 1 and 6, materials must not only comply with IFRA Standards but must also be recognized as safe as a flavoring ingredient as defined by the IOFI Code of Practice (www.iofi.org). For more details see chapter 1 of the Guidance for the use of IFRA Standards. |
| use level in formulae for use in cosmetics: | | | 0.0770 %
|
| Dermal Systemic Exposure in Cosmetic Products: | | | 0.002 mg/kg/day (IFRA, 2002)
|
| |
| Maximised Survey-derived Daily Intakes (MSDI-EU): | 73.00 (μg/capita/day) |
| Maximised Survey-derived Daily Intakes (MSDI-USA): | 5.00 (μg/capita/day) |
| Structure Class: | I |
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). |
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library |
| publication number: 7. Update in publication number(s): 29 |
| Click here to view publication 7 |
| | average usual ppm | average maximum ppm |
| baked goods: | 0.05000 | 0.20000 |
| beverages(nonalcoholic): | 0.05000 | 0.20000 |
| beverages(alcoholic): | 0.01000 | 2.00000 |
| breakfast cereal: | - | - |
| cheese: | - | - |
| chewing gum: | 100.00000 | 300.00000 |
| condiments / relishes: | 0.05000 | 0.20000 |
| confectionery froastings: | - | - |
| egg products: | - | - |
| fats / oils: | - | - |
| fish products: | - | - |
| frozen dairy: | 0.05000 | 0.20000 |
| fruit ices: | - | - |
| gelatins / puddings: | 0.05000 | 0.20000 |
| granulated sugar: | - | - |
| gravies: | - | - |
| hard candy: | 0.20000 | 0.20000 |
| imitation dairy: | - | - |
| instant coffee / tea: | - | - |
| jams / jellies: | 0.05000 | 0.20000 |
| meat products: | - | - |
| milk products: | - | - |
| nut products: | - | - |
| other grains: | - | - |
| poultry: | - | - |
| processed fruits: | - | - |
| processed vegetables: | - | - |
| reconstituted vegetables: | - | - |
| seasonings / flavors: | - | - |
| snack foods: | - | - |
| soft candy: | 0.05000 | 0.20000 |
| soups: | - | - |
| sugar substitutes: | - | - |
| sweet sauces: | - | - |
Safety References:
| Flavor & Extract Manufacturers Association (FEMA) reference(s): |
| The FEMA GRAS assessment of alicyclic substances used as flavor ingredients. View pdf |
| European Food Safety Athority(EFSA): | Flavor usage levels; Subacute, Subchronic, Chronic and Carcinogenicity Studies; Developmental / Reproductive Toxicity Studies; Genotoxicity Studies... |
| European Food Safety Authority (EFSA) reference(s): |
Flavouring Group Evaluation 213: alpha,beta-Unsaturated alicyclic ketones and precursors from chemical subgroup 2.7 of FGE.19
View page or View pdf |
Scientific Opinion on Flavouring Group Evaluation 213, Revision 2 (FGE.213Rev2): Consideration of genotoxic potential for a,�-unsaturated alicyclic ketones and precursors from chemical subgroup 2.7 of FGE.19
View page or View pdf |
Safety and efficacy of secondary alicyclic saturated and unsaturated alcohols, ketones, ketals and esters with ketals containing alicyclic alcohols or ketones and esters containing secondary alicyclic alcohols from chemical group 8 when used as flavourings for all animal species
View page or View pdf |
Safety of 31 flavouring compounds belonging to different chemical groups when used as feed additives for all animal species
View page or View pdf |
| EPI System: | View |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA Substance Registry Services (TSCA): | 23696-85-7 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 5366074 |
| National Institute of Allergy and Infectious Diseases: | Data |
| SCCNFP: | opinion |
| WISER: | UN 1170 |
| WGK Germany: | 2 |
| | (E)-1-(2,6,6-trimethyl-1-cyclohexa-1,3-dienyl)but-2-en-1-one |
| Chemidplus: | 0023696857 |
References:
Other Information:
Potential Blenders and core components note
| |
| For Odor |
| No odor group found for these |
| 3- | mercapto-3-methyl-1-hexanol | |
| 4-(1- | propenyl) pyridine | |
| | satinaldehyde | FL/FR |
| 1-( | thienyl-2)butan-1,2-dione | |
| acidic |
| 2- | methyl-2-pentenoic acid | FL/FR |
| alcoholic |
| | fusel oil | FL/FR |
| aldehydic |
| | decanal (aldehyde C-10) | FL/FR |
| | nonanal (aldehyde C-9) | FL/FR |
| 10- | undecenal (aldehyde C-11 undecylenic) | FL/FR |
| amber |
| | ambrette seed absolute | FL/FR |
| | angelica root oil | FL/FR |
| animal |
| | indole | FL/FR |
| | methyl (E)-2-octenoate | FL/FR |
| 6- | methyl quinoline | FL/FR |
| | skatole | FL/FR |
| anise |
| | anise seed oil colombia | FL/FR |
| sweet | fennel seed oil | FL/FR |
| anisic |
| para- | anisaldehyde | FL/FR |
| | ocimum basilicum herb oil | FL/FR |
| | ocimum basilicum leaf oil america | FL/FR |
| | ocimum basilicum leaf oil CO2 extract | FL/FR |
| aromatic |
| | damascone carboxylate | FR |
| balsamic |
| 2- | acetyl furan | FL/FR |
| iso | amyl benzoate | FL/FR |
| | amyl phenyl acetate | FL/FR |
| | amyris wood oil | FL/FR |
| | benzyl cinnamate | FL/FR |
| iso | butyl cinnamate | FL/FR |
| | cinnamyl alcohol | FL/FR |
| | ethyl cinnamate | FL/FR |
| | fir carboxylate | FR |
| | guaiyl butyrate | FR |
| | terpinyl cinnamate | FL/FR |
| berry |
| | raspberry ketone methyl ether | FL/FR |
| brown |
| sec- | heptyl acetate | FL/FR |
| | ethyl maltol | FL/FR |
| | maltol | FL/FR |
| | strawberry furanone | FL/FR |
| cereal |
| | bran absolute | FR |
| chemical |
| | propyl propionate | FL/FR |
| citrus |
| | grapefruit oil c.p. california | FL/FR |
| | grapefruit pentanol | FR |
| 2- | heptanol | FL/FR |
| blood | orange oil italy | FL/FR |
| 10- | undecen-1-ol | FL/FR |
| creamy |
| 3- | heptyl dihydro-5-methyl-2(3H)-furanone | FL/FR |
| earthy |
| | dibenzyl ether | FL/FR |
| | methyl 3-hexenoate | FL/FR |
| | acetaldehyde dimethyl acetal | FL/FR |
| | cyclohexyl formate | FL/FR |
| 1- | hexen-3-ol | FL/FR |
| | methyl ethyl ketone | FL/FR |
| 2- | methyl valeraldehyde | FL/FR |
| | propyl formate | FL/FR |
| fatty |
| 3- | decen-2-one | FL/FR |
| (E,Z)-2,6- | dodecadienal | FL/FR |
| | ethyl undecylenate | FL/FR |
| | hexyl pivalate | FR |
| floral |
| | allyl anthranilate | FL/FR |
| alpha- | amyl cinnamyl acetate | FL/FR |
| iso | amyl salicylate | FL/FR |
| | anise indene | FR |
| para- | anisyl butyrate | FL/FR |
| | benzyl acetate | FL/FR |
| | bois de rose oil brazil | FL/FR |
| | bois de rose oil peru | FL/FR |
| | boronia absolute | FL/FR |
| | boronia butenal | FR |
| iso | butyl salicylate | FL/FR |
| | cardamom absolute | FL/FR |
| | cassie absolute | FL/FR |
| | cassis buteneone | FR |
| | citronellal diisotridecyl acetal | FR |
| | citronellol | FL/FR |
| | citronellyl acetate | FL/FR |
| | citronellyl anthranilate | FL/FR |
| | citronellyl formate | FL/FR |
| | citronellyl hexanoate | FL/FR |
| | citronellyl propionate | FL/FR |
| (E)- | citronellyl tiglate | FL/FR |
| | coriander seed oil | FL/FR |
| para- | cresyl acetate | FL/FR |
| | cuminyl acetaldehyde | FL/FR |
| | cyclamen aldehyde | FL/FR |
| 3- | cyclohexene-1-carboxylic acid, 2,6,6-trimethyl-, methyl ester | FR |
| | cyclohexyl ethyl alcohol | FL/FR |
| delta- | damascone | FL/FR |
| | dihydro-alpha-ionone | FL/FR |
| | dihydrojasmone | FL/FR |
| | dimethyl anthranilate | FL/FR |
| | dimethyl benzyl carbinyl acetate | FL/FR |
| | dimethyl benzyl carbinyl butyrate | FL/FR |
| 2,4- | dimethyl-alpha-allyl-3-cyclohexene methanol | |
| 4,6- | dimethyl-alpha-allyl-3-cyclohexene methanol | |
| | ethyl ortho-anisate | FL/FR |
| | eucalyptus macarthurii oil | FR |
| | floral pyranol | FR |
| | geraniol | FL/FR |
| | geranium oil egypt fractions | FR |
| | geranyl acetate | FL/FR |
| (E)- | geranyl acetone | FL/FR |
| | geranyl formate | FL/FR |
| | geranyl phenyl acetate | FL/FR |
| | geranyl valerate | FL/FR |
| | heliotropin | FL/FR |
| | heliotropyl acetone | FL/FR |
| (Z)-3- | hexen-1-yl salicylate | FL/FR |
| alpha- | hexyl cinnamaldehyde | FL/FR |
| | hyacinth acetals | FL/FR |
| beta- | ionone | FL/FR |
| (E)-beta- | ionone | FL/FR |
| beta- | ionyl acetate | FL/FR |
| iso | jasmone | FL/FR |
| iso | jasmone | FL/FR |
| | leerall | FR |
| | lilac pentanol | FL/FR |
| | linalool | FL/FR |
| | linalool oxide | FL/FR |
| | linalyl propionate | FL/FR |
| | methyl dihydrojasmonate | FL/FR |
| (Z)- | methyl epi-jasmonate | FL/FR |
| N- | methyl ionone | FR |
| alpha-iso | methyl ionone (90% min.) | FL/FR |
| | methyl jasmonate | FL/FR |
| | mimosa absolute | FL/FR |
| | mimosa absolute france | FL/FR |
| | mimosa absolute india | FL/FR |
| | muguet octadienol | FR |
| | nerol | FL/FR |
| | nerolidol | FL/FR |
| | neryl acetate | FL/FR |
| | neryl formate | FL/FR |
| | ocean propanal | FL/FR |
| beta- | ocimene | FL/FR |
| (Z)-beta- | ocimene | FL/FR |
| | peony alcohol | FR |
| | petitgrain bigarade oil | FL/FR |
| | petitgrain lemon oil | FL/FR |
| | petitgrain mandarin oil | FL/FR |
| | petitgrain oil paraguay | FL/FR |
| | phenethyl acetate | FL/FR |
| | phenethyl isobutyrate | FL/FR |
| | phenethyl isovalerate | FL/FR |
| 2- | phenethyl valerate | FL/FR |
| 2- | phenyl propionaldehyde dimethyl acetal | FL/FR |
| 1- | phenyl propyl butyrate | FL/FR |
| | plum damascone | FR |
| | propyl salicylate | FR |
| | rose butanoate | FL/FR |
| | styralyl propionate | FL/FR |
| | tobacco flower absolute | FR |
| | violet methyl carbonate | FR |
| | ylang ylang flower oil | FL/FR |
| fresh |
| 10- | undecen-1-yl acetate | FL/FR |
| fruity |
| | allyl amyl glycolate | FR |
| | allyl butyrate | FL/FR |
| | allyl cyclohexyl propionate | FL/FR |
| | allyl isononylate | FR |
| alpha- | amyl cinnamyl isovalerate | FL/FR |
| | amyl formate | FL/FR |
| iso | amyl hexanoate | FL/FR |
| iso | amyl isobutyrate | FL/FR |
| iso | amyl isovalerate | FL/FR |
| | apple crotonate | FR |
| | apricot isobutyrate | FR |
| | benzyl propionate | FL/FR |
| 3- | benzyl-4-heptanone | FL/FR |
| | berry pentadienoate | FL/FR |
| | bisabolene | FL/FR |
| | butyl 2-decenoate | FL/FR |
| | butyl anthranilate | FL/FR |
| | butyl formate | FL/FR |
| | butyl hexanoate | FL/FR |
| iso | butyl isovalerate | FL/FR |
| | cherry pentenoate | FL/FR |
| | citronellyl isobutyrate | FL/FR |
| | cyclohexanone diethyl acetal | FL/FR |
| (E)-beta- | damascone | FL/FR |
| (Z)-beta- | damascone | FL/FR |
| beta- | damascone | FL/FR |
| gamma- | decalactone | FL/FR |
| alpha,alpha- | dimethyl benzyl isobutyrate | FL/FR |
| | dimethyl succinate | FL/FR |
| | ethyl 2-octenoate | FL/FR |
| | ethyl 3-hexenoate | FL/FR |
| | ethyl 3-hydroxyhexanoate | FL/FR |
| | ethyl 4-phenyl butyrate | FL/FR |
| | ethyl acetoacetate | FL/FR |
| | ethyl heptanoate | FL/FR |
| | ethyl isovalerate | FL/FR |
| | ethyl levulinate | FL/FR |
| (E)- | ethyl tiglate | FL/FR |
| (E,Z)-6- | ethyl-5,7-dimethyl-2,5-octadien-4-one | FR |
| | geranyl butyrate | FL/FR |
| | geranyl isovalerate | FL/FR |
| | grape butyrate | FL/FR |
| | green acetate | FR |
| | hexanal propylene glycol acetal | FL/FR |
| 2- | hexen-1-ol | FL/FR |
| (E)-3- | hexen-1-yl acetate | FL/FR |
| | hexyl acetate | FL/FR |
| | hexyl formate | FL/FR |
| | hexyl isovalerate | FL/FR |
| | linalool oxide acetates | FL/FR |
| | methyl formyl anthranilate | FL/FR |
| | methyl heptanoate | FL/FR |
| 3- | methyl-2-butenal | FL/FR |
| | neryl propionate | FL/FR |
| 2- | nonanone | FL/FR |
| | octyl butyrate | FL/FR |
| | octyl formate | FL/FR |
| 3- | phenyl propyl isovalerate | FL/FR |
| | plum crotonate | FR |
| | plum damascone (high alpha) | FR |
| (E,E)- | pomarose | FR |
| | prenol | FL/FR |
| | propyl angelate | |
| | strawberry glycidate 1 (aldehyde C-16 (so-called)) | FL/FR |
| | thesaron (Takasago) | FR |
| para- | tolualdehyde | FL/FR |
| 4-(para- | tolyl)-2-butanone | FL/FR |
| gamma- | undecalactone (aldehyde C-14 (so-called)) | FL/FR |
| fungal |
| | methyl 2-furoate | FL/FR |
| green |
| | actinidia chinensis fruit extract | FL/FR |
| iso | amyl formate | FL/FR |
| | ethyl (E,Z)-2,4-decadienoate | FL/FR |
| | ethyl (E)-2-hexenoate | FL/FR |
| | galbanum oil | FL/FR |
| | heptyl formate | FL/FR |
| (E)-3- | hexen-1-ol | FL/FR |
| (Z)-3- | hexen-1-yl (Z)-3-hexenoate | FL/FR |
| (E)-2- | hexen-1-yl acetate | FL/FR |
| (Z)-3- | hexen-1-yl acetate | FL/FR |
| (Z)-3- | hexen-1-yl benzoate | FL/FR |
| (Z)-3- | hexen-1-yl butyrate | FL/FR |
| (Z)-3- | hexen-1-yl hexanoate | FL/FR |
| (Z)-3- | hexen-1-yl isovalerate | FL/FR |
| (E)-2- | hexen-1-yl isovalerate | FL/FR |
| (Z)-3- | hexen-1-yl propionate | FL/FR |
| (Z)-3- | hexen-1-yl tiglate | FL/FR |
| (E)-2- | hexen-1-yl valerate | FL/FR |
| 3- | hexenyl 2-methyl butyrate | FL/FR |
| (Z)-3- | hexenyl methyl ether | FR |
| | hexyl hexanoate | FL/FR |
| | hexyl phenyl acetate | FL/FR |
| | hexyl tiglate | FL/FR |
| (Z)- | leaf acetal | FL/FR |
| | melon nonenoate | FL/FR |
| | methyl (E)-3-hexenoate | FL/FR |
| | methyl heptine carbonate | FL/FR |
| | methyl octine carbonate | FL/FR |
| | neryl butyrate | FL/FR |
| (E,Z)-2,6- | nonadien-1-ol | FL/FR |
| 3- | octyl formate | FL/FR |
| (E)-2- | pentenal | FL/FR |
| | phenoxyethyl isobutyrate | FL/FR |
| 2- | phenyl propionaldehyde | FL/FR |
| 1- | phenyl-2-pentanol | FL/FR |
| (E,E)- | sorbyl acetate | |
| | terpinyl propionate | FL/FR |
| | thiogeraniol | FL/FR |
| | tiglaldehyde | FL/FR |
| | violet leaf absolute | FL/FR |
| hay |
| | beeswax absolute | FL/FR |
| | hay absolute | FR |
| | tobacco leaf absolute | FL/FR |
| | woodruff absolute | FR |
| herbal |
| | anthemis nobilis flower extract | FL/FR |
| | anthemis nobilis flower oil roman | FL/FR |
| sweet | basil absolute | FL/FR |
| (S)- | campholene acetate | FL/FR |
| | carum carvi fruit oil | FL/FR |
| 1,4- | cineole | FL/FR |
| | clary sage oil france | FL/FR |
| | coriander oleoresin | FL/FR |
| | geranium cyclohexane | FR |
| | geranyl octanoate | FL/FR |
| | herbal undecanone | FR |
| | hexanol | FL/FR |
| | hyssop oil | FL/FR |
| | linalyl formate | FL/FR |
| | linalyl octanoate | FL/FR |
| | matricaria chamomilla flower oil | FL/FR |
| | methyl cyclogeranate (Firmenich) | FR |
| | methyl nicotinate | FL/FR |
| | nonisyl acetate | FR |
| curled | parsley seed oil | FL/FR |
| beta-sesqui | phellandrene | |
| | rosmarinus officinalis extract | FL/FR |
| | rosmarinus officinalis tincture | FL/FR |
| | saffron indenone | FL/FR |
| | safranal | FL/FR |
| | salvia sclarea oil | FL/FR |
| | terpinolene | FL/FR |
| | theaspirane | FL/FR |
| | yerba mate absolute | FL/FR |
| honey |
| | phenyl acetic acid | FL/FR |
| melon |
| | melon heptenal | FL/FR |
| (Z)-6- | nonenal | FL/FR |
| | watermelon ketone | FR |
| minty |
| (-)- | menthone | FL/FR |
| mossy |
| | oakmoss absolute | FL/FR |
| | veramoss (IFF) | FR |
| musty |
| ketoiso | phorone | FL/FR |
| phenolic |
| 2'- | hydroxyacetophenone | FL/FR |
| popcorn |
| 2- | acetyl pyridine | FL/FR |
| powdery |
| para- | anisyl acetate | FL/FR |
| para- | anisyl alcohol | FL/FR |
| (E)-alpha- | methyl ionone (74-80%) | FL/FR |
| spicy |
| para- | anisyl formate | FL/FR |
| | cassia bark oil china | FL/FR |
| | clove bud oil | FL/FR |
| | cuminaldehyde | FL/FR |
| black | currant bud absolute | FL/FR |
| N,N- | diethyl octanamide | FR |
| iso | eugenyl acetate | FL/FR |
| terpenic |
| | cassis bud oil | FL/FR |
| alpha- | terpineol | FL/FR |
| tobacco |
| para- | cresyl isovalerate | FL/FR |
| 3- | ethyl pyridine | FL/FR |
| | methyl benzoxole | FL/FR |
| | tobacco concrete | FR |
| (E,E/E,Z)- | tobacco cyclohexenone | FL/FR |
| | veltonal (Bedoukian) | FR |
| tonka |
| | deertongue absolute | FR |
| gamma- | hexalactone | FL/FR |
| | mint lactone | FL/FR |
| | saffron resinoid | FL/FR |
| | tonka undecanone | FR |
| tropical |
| | genet absolute | FL/FR |
| | hotrienol | FL/FR |
| | psidium guajava fruit extract | FL/FR |
| vanilla |
| | propenyl guaethol | FL/FR |
| | vanilla bean absolute (vanilla planifolia) | FL/FR |
| | vanillyl acetate | FL/FR |
| | methyl octanoate | FL/FR |
| 2- | nonanol | FL/FR |
| winey |
| | butyl angelate | FL/FR |
| woody |
| | amber decatriene | FR |
| alpha- | cedrene epoxide | FR |
| | cistus twig/leaf oil | FL/FR |
| 2- | decalinyl acetate | FR |
| 3',4'- | dimethoxyacetophenone | |
| iso | longifolene ketone | FR |
| | myrtenyl isobutyrate | |
| | patchouli absolute | FR |
| | santall | FR |
| | santalyl butyrate | FL/FR |
| | spicy pentanone | FL/FR |
| | tobacarol (IFF) | FR |
| | tobacco nonene | FR |
| (Z)- | woody amylene | FR |
| | woody dioxolane | FR |
| | woody ether | FR |
| |
| For Flavor |
| |
| No flavor group found for these |
| | allyl anthranilate | FL/FR |
| | amyl phenyl acetate | FL/FR |
| | butyl angelate | FL/FR |
| | butyl formate | FL/FR |
| | capsicum oleoresin | FL |
| | cyclohexanone diethyl acetal | FL/FR |
| | ethyl 4-phenyl butyrate | FL/FR |
| | ethyl ortho-anisate | FL/FR |
| | fig leaf absolute | FL |
| | geranyl octanoate | FL/FR |
| | geranyl valerate | FL/FR |
| 2- | hexenal | FL |
| | linalool oxide acetates | FL/FR |
| | methyl formyl anthranilate | FL/FR |
| | methyl nicotinate | FL/FR |
| | myrtenyl isobutyrate | |
| beta-sesqui | phellandrene | |
| 2- | phenethyl valerate | FL/FR |
| 1- | phenyl propyl butyrate | FL/FR |
| 3- | phenyl propyl isovalerate | FL/FR |
| | propyl angelate | |
| 2- | propyl pyridine | FL |
| | santalyl butyrate | FL/FR |
| | skatole | FL/FR |
| 10- | undecen-1-ol | FL/FR |
|
| beta- | damascone | FL/FR |
| absinthe |
| | absinthe flavor | FL |
| aldehydic |
| | nonanal (aldehyde C-9) | FL/FR |
| animal |
| | indole | FL/FR |
| 6- | methyl quinoline | FL/FR |
| anise |
| | anise seed oil colombia | FL/FR |
| sweet | fennel seed oil | FL/FR |
| anisic |
| para- | anisyl butyrate | FL/FR |
| apple |
| (E,Z)-2,6- | nonadien-1-ol | FL/FR |
| aromatic |
| para- | cresyl acetate | FL/FR |
| | hyacinth acetals | FL/FR |
| astringent |
| 1-( | thienyl-2)butan-1,2-dione | |
| balsamic |
| iso | butyl cinnamate | FL/FR |
| | ethyl cinnamate | FL/FR |
| berry |
| | dihydro-alpha-ionone | FL/FR |
| | heliotropyl acetone | FL/FR |
| | raspberry ketone methyl ether | FL/FR |
| brown |
| | beeswax absolute | FL/FR |
| caramellic |
| | caramel furanone | FL |
| | ethyl maltol | FL/FR |
| 3- | ethyl pyridine | FL/FR |
| | maltol | FL/FR |
| | methyl 2-furoate | FL/FR |
| | strawberry furanone | FL/FR |
| cheesy |
| 2- | nonanone | FL/FR |
| chemical |
| | methyl ethyl ketone | FL/FR |
| cherry |
| | heliotropin | FL/FR |
| citrus |
| | bisabolene | FL/FR |
| | grapefruit oil c.p. california | FL/FR |
| | linalool | FL/FR |
| | nerol | FL/FR |
| blood | orange oil italy | FL/FR |
| | petitgrain bigarade oil | FL/FR |
| | petitgrain lemon oil | FL/FR |
| | petitgrain mandarin oil | FL/FR |
| ketoiso | phorone | FL/FR |
| | styralyl propionate | FL/FR |
| alpha- | terpineol | FL/FR |
| coconut |
| (R)- | massoia lactone | FL |
| cooling |
| iso | butyl salicylate | FL/FR |
| 1,4- | cineole | FL/FR |
| | theaspirane | FL/FR |
| corn |
| 2- | acetyl pyridine | FL/FR |
| creamy |
| para- | anisaldehyde | FL/FR |
| gamma- | hexalactone | FL/FR |
| | massoia lactone | FL |
| | mint lactone | FL/FR |
| gamma- | undecalactone (aldehyde C-14 (so-called)) | FL/FR |
| earthy |
| alpha- | amyl cinnamyl acetate | FL/FR |
| estery |
| | ethyl acetoacetate | FL/FR |
| ethereal |
| | acetaldehyde dimethyl acetal | FL/FR |
| fatty |
| | ethyl undecylenate | FL/FR |
| sec- | heptyl acetate | FL/FR |
| | heptyl formate | FL/FR |
| (Z)-3- | hexen-1-yl benzoate | FL/FR |
| 10- | undecenal (aldehyde C-11 undecylenic) | FL/FR |
| floral |
| | bois de rose oil brazil | FL/FR |
| | bois de rose oil peru | FL/FR |
| | cardamom absolute | FL/FR |
| | citronellol | FL/FR |
| | citronellyl acetate | FL/FR |
| | citronellyl hexanoate | FL/FR |
| | citronellyl propionate | FL/FR |
| | dihydrojasmone | FL/FR |
| | dimethyl benzyl carbinyl acetate | FL/FR |
| | dimethyl benzyl carbinyl butyrate | FL/FR |
| 2,4- | dimethyl-alpha-allyl-3-cyclohexene methanol | |
| 4,6- | dimethyl-alpha-allyl-3-cyclohexene methanol | |
| | geraniol | FL/FR |
| (E)- | geranyl acetone | FL/FR |
| | geranyl phenyl acetate | FL/FR |
| | hotrienol | FL/FR |
| | methyl dihydrojasmonate | FL/FR |
| (Z)- | methyl epi-jasmonate | FL/FR |
| (E)-alpha- | methyl ionone (74-80%) | FL/FR |
| alpha-iso | methyl ionone (90% min.) | FL/FR |
| | methyl jasmonate | FL/FR |
| | neryl acetate | FL/FR |
| | ocean propanal | FL/FR |
| | phenyl acetic acid | FL/FR |
| | satinaldehyde | FL/FR |
| | ylang ylang flower oil | FL/FR |
| fruity |
| | allyl cyclohexyl propionate | FL/FR |
| iso | amyl benzoate | FL/FR |
| alpha- | amyl cinnamyl isovalerate | FL/FR |
| | amyl formate | FL/FR |
| iso | amyl hexanoate | FL/FR |
| iso | amyl isobutyrate | FL/FR |
| para- | anisyl acetate | FL/FR |
| para- | anisyl alcohol | FL/FR |
| | benzyl acetate | FL/FR |
| | benzyl propionate | FL/FR |
| 3- | benzyl-4-heptanone | FL/FR |
| | berry pentadienoate | FL/FR |
| | boronia absolute | FL/FR |
| | butyl 2-decenoate | FL/FR |
| | butyl anthranilate | FL/FR |
| | butyl hexanoate | FL/FR |
| | cassis bud oil | FL/FR |
| | cherry pentenoate | FL/FR |
| | citronellyl anthranilate | FL/FR |
| | citronellyl formate | FL/FR |
| | citronellyl isobutyrate | FL/FR |
| (Z)-beta- | damascone | FL/FR |
| (E)-beta- | damascone | FL/FR |
| gamma- | decalactone | FL/FR |
| | dibenzyl ether | FL/FR |
| | dimethyl anthranilate | FL/FR |
| alpha,alpha- | dimethyl benzyl isobutyrate | FL/FR |
| | dimethyl succinate | FL/FR |
| | ethyl (E)-2-hexenoate | FL/FR |
| | ethyl (E)-2-octenoate | FL |
| | ethyl 2-octenoate | FL/FR |
| | ethyl 3-hexenoate | FL/FR |
| | ethyl 3-hydroxyhexanoate | FL/FR |
| | ethyl heptanoate | FL/FR |
| | ethyl isovalerate | FL/FR |
| | ethyl levulinate | FL/FR |
| (E)- | ethyl tiglate | FL/FR |
| | furfuryl isovalerate | FL |
| 2- | furyl pentyl ketone | FL |
| | fusel oil | FL/FR |
| | geranyl butyrate | FL/FR |
| 2- | heptanol | FL/FR |
| 3- | heptyl dihydro-5-methyl-2(3H)-furanone | FL/FR |
| | hexanal propylene glycol acetal | FL/FR |
| 2- | hexen-1-ol | FL/FR |
| (E)-3- | hexen-1-yl acetate | FL/FR |
| | hexyl acetate | FL/FR |
| | hexyl hexanoate | FL/FR |
| | hexyl phenyl acetate | FL/FR |
| | kiwi distillates | FL |
| | lilac pentanol | FL/FR |
| | linalyl octanoate | FL/FR |
| 3- | mercapto-3-methyl-1-hexanol | |
| | methyl (E)-2-octenoate | FL/FR |
| | methyl 3-hexenoate | FL/FR |
| | methyl formate | FL |
| | methyl heptanoate | FL/FR |
| 3- | methyl-2-butenal | FL/FR |
| | neryl formate | FL/FR |
| (Z)-3- | nonen-1-yl acetate | FL |
| (Z)-5- | octen-1-yl acetate | FL |
| | phenethyl isovalerate | FL/FR |
| 2- | phenyl propionaldehyde dimethyl acetal | FL/FR |
| 1- | phenyl-2-pentanol | FL/FR |
| | prenol | FL/FR |
| | propyl formate | FL/FR |
| | rose butanoate | FL/FR |
| | spicy pentanone | FL/FR |
| | strawberry glycidate 1 (aldehyde C-16 (so-called)) | FL/FR |
| | terpinyl cinnamate | FL/FR |
| | tiglaldehyde | FL/FR |
| 4-(para- | tolyl)-2-butanone | FL/FR |
| grassy |
| | tobacco leaf absolute | FL/FR |
| greasy |
| 10- | undecen-1-yl acetate | FL/FR |
| green |
| | actinidia chinensis fruit extract | FL/FR |
| | allyl butyrate | FL/FR |
| iso | amyl formate | FL/FR |
| iso | amyl isovalerate | FL/FR |
| iso | amyl salicylate | FL/FR |
| | angelica root oil | FL/FR |
| iso | butyl isovalerate | FL/FR |
| | cinnamyl alcohol | FL/FR |
| (E)- | citronellyl tiglate | FL/FR |
| | cuminyl acetaldehyde | FL/FR |
| | cyclamen aldehyde | FL/FR |
| | cyclohexyl ethyl alcohol | FL/FR |
| | cyclohexyl formate | FL/FR |
| 3- | decen-2-one | FL/FR |
| (E,Z)-2,6- | dodecadienal | FL/FR |
| | ethyl (E,Z)-2,4-decadienoate | FL/FR |
| 2- | ethyl butyraldehyde | FL |
| | galbanum oil | FL/FR |
| | geranyl acetate | FL/FR |
| | geranyl formate | FL/FR |
| | geranyl isovalerate | FL/FR |
| | grape butyrate | FL/FR |
| | hexanol | FL/FR |
| (E)-3- | hexen-1-ol | FL/FR |
| (Z)-3- | hexen-1-yl (Z)-3-hexenoate | FL/FR |
| (E)-2- | hexen-1-yl acetate | FL/FR |
| (Z)-3- | hexen-1-yl acetate | FL/FR |
| (Z)-3- | hexen-1-yl butyrate | FL/FR |
| (Z)-3- | hexen-1-yl hexanoate | FL/FR |
| (E)-2- | hexen-1-yl isovalerate | FL/FR |
| (Z)-3- | hexen-1-yl isovalerate | FL/FR |
| (Z)-3- | hexen-1-yl propionate | FL/FR |
| (Z)-3- | hexen-1-yl salicylate | FL/FR |
| (Z)-3- | hexen-1-yl tiglate | FL/FR |
| (E)-2- | hexen-1-yl valerate | FL/FR |
| 1- | hexen-3-ol | FL/FR |
| 3- | hexenyl 2-methyl butyrate | FL/FR |
| | hexyl formate | FL/FR |
| | hexyl isovalerate | FL/FR |
| | hexyl tiglate | FL/FR |
| iso | jasmone | FL/FR |
| iso | jasmone | FL/FR |
| (Z)- | leaf acetal | FL/FR |
| | linalool oxide | FL/FR |
| | melon heptenal | FL/FR |
| | melon nonenoate | FL/FR |
| | methyl (E)-3-hexenoate | FL/FR |
| | methyl 2-undecynoate | FL |
| | methyl heptine carbonate | FL/FR |
| | methyl octanoate | FL/FR |
| | methyl octine carbonate | FL/FR |
| 3-(5- | methyl-2-furyl) butanal | FL |
| 4- | methyl-2-pentenal | FL |
| | nerolidol | FL/FR |
| | neryl butyrate | FL/FR |
| | neryl propionate | FL/FR |
| (Z)-6- | nonenal | FL/FR |
| | oakmoss absolute | FL/FR |
| beta- | ocimene | FL/FR |
| (Z)-beta- | ocimene | FL/FR |
| (E,E)-2,4- | octadienal | FL |
| 2,4- | octadienal | FL |
| | octyl formate | FL/FR |
| (E)-2- | pentenal | FL/FR |
| | phenoxyethyl isobutyrate | FL/FR |
| 2- | phenyl propionaldehyde | FL/FR |
| 4-(1- | propenyl) pyridine | |
| (E,E)- | sorbyl acetate | |
| | sorbyl acetate | FL |
| | terpinyl propionate | FL/FR |
| | thiogeraniol | FL/FR |
| | violet leaf absolute | FL/FR |
| hay |
| | genet absolute | FL/FR |
| herbal |
| | anthemis nobilis flower extract | FL/FR |
| | anthemis nobilis flower oil roman | FL/FR |
| sweet | basil absolute | FL/FR |
| | carum carvi fruit oil | FL/FR |
| | clary sage oil france | FL/FR |
| | coriander oleoresin | FL/FR |
| | coriander seed oil | FL/FR |
| | hyssop oil | FL/FR |
| | linalyl formate | FL/FR |
| | linalyl propionate | FL/FR |
| | matricaria chamomilla flower oil | FL/FR |
| | ocimum basilicum herb oil | FL/FR |
| | ocimum basilicum leaf oil america | FL/FR |
| | ocimum basilicum leaf oil CO2 extract | FL/FR |
| curled | parsley seed oil | FL/FR |
| | petitgrain oil paraguay | FL/FR |
| | rosmarinus officinalis extract | FL/FR |
| | rosmarinus officinalis tincture | FL/FR |
| | saffron indenone | FL/FR |
| | salvia sclarea oil | FL/FR |
| | yerba mate absolute | FL/FR |
| honey |
| | phenethyl acetate | FL/FR |
| | phenethyl isobutyrate | FL/FR |
| jammy |
| (S)- | campholene acetate | FL/FR |
| minty |
| (-)- | menthone | FL/FR |
| naphthyl |
| 2'- | hydroxyacetophenone | FL/FR |
| nutty |
| 2- | acetyl furan | FL/FR |
| | methyl benzoxole | FL/FR |
| (E,E/E,Z)- | tobacco cyclohexenone | FL/FR |
| phenolic |
| para- | cresyl isovalerate | FL/FR |
| pungent |
| | acetaldehyde | FL |
| sour |
| 2- | methyl-2-pentenoic acid | FL/FR |
| spicy |
| para- | anisyl formate | FL/FR |
| | benzyl cinnamate | FL/FR |
| | cassia bark oil china | FL/FR |
| | cassie absolute | FL/FR |
| | clove bud oil | FL/FR |
| | cuminaldehyde | FL/FR |
| black | currant bud absolute | FL/FR |
| iso | eugenyl acetate | FL/FR |
| | paprika oleoresin | FL |
| para- | tolualdehyde | FL/FR |
| sweet |
| | saffron resinoid | FL/FR |
| tea |
| red rooibos | tea leaf distillates | FL |
| tropical |
| | guava distillates | FL |
| | propyl propionate | FL/FR |
| | psidium guajava fruit | FL |
| | psidium guajava fruit extract | FL/FR |
| vanilla |
| | propenyl guaethol | FL/FR |
| | vanilla bean absolute (vanilla planifolia) | FL/FR |
| | vanillyl acetate | FL/FR |
| vegetable |
| 2- | methyl valeraldehyde | FL/FR |
| waxy |
| | decanal (aldehyde C-10) | FL/FR |
| alpha- | hexyl cinnamaldehyde | FL/FR |
| | mimosa absolute | FL/FR |
| | mimosa absolute france | FL/FR |
| | mimosa absolute india | FL/FR |
| 2- | nonanol | FL/FR |
| | octyl butyrate | FL/FR |
| 3- | octyl formate | FL/FR |
| woody |
| | ambrette seed absolute | FL/FR |
| | amyris wood oil | FL/FR |
| | cistus twig/leaf oil | FL/FR |
| delta- | damascone | FL/FR |
| 3',4'- | dimethoxyacetophenone | |
| beta- | ionone | FL/FR |
| (E)-beta- | ionone | FL/FR |
| beta- | ionyl acetate | FL/FR |
| | safranal | FL/FR |
| | terpinolene | FL/FR |
| |
Potential Uses:
Occurrence (nature, food, other): noteSynonyms:
| 2- | buten-1-one, 1-(2,6,6-trimethyl-1,3-cyclohexadien-1-yl)-, (2E)- | | 2- | buten-1-one, 1-(2,6,6-trimethyl-1,3-cyclohexadien-1-yl)-, (E)- | | | damascenone | | beta- | damascenone | | beta- | damascenone 1,5% in ethanol natural | | | damascenone 1% in ETOH natural | | | damascenone 1% in PG natural | | | damascenone beta at 1% in ethanol | | | damascenone beta at 1% in PG | | | damascenone beta key 100 | | | damascenone beta key 500 | | | damascenone beta key 500 halal in PG | | | damascenone natural | | beta- | damascenone natural | | | damascenone pure | | | fermentone (Advanced Biotech) | | | fermentone 1% in PG natural (Advanced Biotech) | | | fermentone 5% ETOH natural (Advanced Biotech) | | | fermentone extra 1% in triacetin natural (Advanced Biotech) | | | fermentone extra natural (Advanced Biotech) | | | fermentone natural | | | floriffone | | | roastarome natural (Advanced Biotech) | | | rose ketone-4 | | | rosenone | | 4-(2,6,6- | trimethyl cyclohexa-1,3-dienyl) but-2-en-4-one | | | trimethyl cyclohexadienyl butenone | | trans-2,6,6- | trimethyl-1-(2-butenoyl)cyclohexa-1,3-diene | | (E)-1-(2,6,6- | trimethyl-1-cyclohexa-1,3-dienyl)but-2-en-1-one | | 2,6,6- | trimethyl-1-trans-crotonoyl-1,3-cyclohexadiene | | (2,6,6- | trimethyl-1,3-cyclohexadien-1-yl) butenone | | (2E)-1-(2,6,6- | trimethyl-1,3-cyclohexadien-1-yl)-2-buten-1-one | | (E)-1-(2,6,6- | trimethyl-1,3-cyclohexadien-1-yl)-2-buten-1-one | | 1-(2,6,6- | trimethyl-1,3-cyclohexadien-1-yl)-2-buten-1-one | | (2E)-1-(2,6,6- | trimethylcyclohexa-1,3-dien-1-yl)but-2-en-1-one |
Articles:
| Google Patents: | Dimethyl sulfide, beta-damascenone, 3-methylbutanal, and 3-methylbutyric acid |
| US Patents: | 3,959,508 - Flavoring compositions containing mixture of 2,2,6-trimethyl-1-cyclohexen-1-ylacetaldehyde and 2,6,6-trimethyl-1-crotonyl-1,3-cyclohexadiene |
| PubMed: | Characterization of the key aroma compounds in Shiraz wine by quantitation, aroma reconstitution, and omission studies. |
| PubMed: | Identification of Potent Odorants in a Novel Nonalcoholic Beverage Produced by Fermentation of Wort with Shiitake (Lentinula edodes). |
| PubMed: | Chemical composition of the essential oil of Croton bonplandianus from India. |
| PubMed: | Influence of pre-fermentation cold maceration treatment on aroma compounds of Cabernet Sauvignon wines fermented in different industrial scale fermenters. |
| PubMed: | Comparison of aroma active and sulfur volatiles in three fragrant rice cultivars using GC-olfactometry and GC-PFPD. |
| PubMed: | Pinot Noir wine composition from different vine vigour zones classified by remote imaging technology. |
| PubMed: | Volatile constituents of commercial imported and domestic black-ripe table olives (Olea europaea). |
| PubMed: | Characterization of the key aroma compounds in two bavarian wheat beers by means of the sensomics approach. |
| PubMed: | Flavor of cold-hardy grapes: impact of berry maturity and environmental conditions. |
| PubMed: | Phytochemical and physical-chemical analysis of Polish willow (Salix spp.) honey: identification of the marker compounds. |
| PubMed: | Characterization of the key aroma compounds in Bartlett pear brandies by means of the sensomics concept. |
| PubMed: | Cooked carrot volatiles. AEDA and odor activity comparisons. Identification of linden ether as an important aroma component. |
| PubMed: | Characterization of aroma-active compounds in dry flower of Malva sylvestris L. by GC-MS-O analysis and OAV calculations. |
| PubMed: | Identification of regions associated with variation in sensitivity to food-related odors in the human genome. |
| PubMed: | Characterization of Muscat wines aroma evolution using comprehensive gas chromatography followed by a post-analytic approach to 2D contour plots comparison. |
| PubMed: | Essential oils from the leaves of six medicinal plants of Nigeria. |
| PubMed: | Accelerated aging against conventional storage: effects on the volatile composition of chardonnay white wines. |
| PubMed: | Key odorants in cured Madagascar vanilla beans (Vanilla planiforia) of differing bean quality. |
| PubMed: | Identification of the key odorants in Tahitian cured vanilla beans (Vanilla tahitensis) by GC-MS and an aroma extract dilution analysis. |
| PubMed: | Sonication in combination with heat and low pressure as an alternative pasteurization treatment--effect on Escherichia coli K12 inactivation and quality of apple cider. |
| PubMed: | Changes in the bound aroma profiles of 'Hayward' and 'Hort16A' kiwifruit (Actinidia spp.) during ripening and GC-olfactometry analysis. |
| PubMed: | Influence of deficit irrigation and kaolin particle film on grape composition and volatile compounds in Merlot grape (Vitis vinifera L.). |
| PubMed: | System design for integrated comprehensive and multidimensional gas chromatography with mass spectrometry and olfactometry. |
| PubMed: | A novel mechanism of filaggrin induction and sunburn prevention by β-damascenone in Skh-1 mice. |
| PubMed: | Quantification by solid phase micro extraction and stable isotope dilution assay of norisoprenoid compounds in red wines obtained from Piedmont rare varieties. |
| PubMed: | Environmental and seasonal influences on red raspberry flavour volatiles and identification of quantitative trait loci (QTL) and candidate genes. |
| PubMed: | Evaluation of key odorants in sauvignon blanc wines using three different methodologies. |
| PubMed: | Characterization of the key aroma compounds in rape honey by means of the molecular sensory science concept. |
| PubMed: | The chemical composition of the essential oils of Cirsium palustre and C. rivulare and their antiproliferative effect. |
| PubMed: | Characterization of volatiles and aroma-active compounds in honeybush (Cyclopia subternata) by GC-MS and GC-O analysis. |
| PubMed: | Odor potency of aroma compounds in Riesling and Vidal blanc table wines and icewines by gas chromatography-olfactometry-mass spectrometry. |
| PubMed: | Experience influences elemental and configural perception of certain binary odour mixtures in newborn rabbits. |
| PubMed: | Volatile compounds responsible for aroma of Jutrzenka liquer wine. |
| PubMed: | Impact of cover crops in vineyard on the aroma compounds of Vitis vinifera L. cv Cabernet Sauvignon wine. |
| PubMed: | Quality and aromatic sensory descriptors (mainly fresh and dry fruit character) of Spanish red wines can be predicted from their aroma-active chemical composition. |
| PubMed: | Characterization of odor-active compounds in guava wine. |
| PubMed: | Chemical characterization of commercial Sherry vinegar aroma by headspace solid-phase microextraction and gas chromatography-olfactometry. |
| PubMed: | Hyperoxygenation and bottle storage of Chardonnay white wines: effects on color-related phenolics, volatile composition, and sensory characteristics. |
| PubMed: | Antioxidant activity and chemical composition of essential oil from Atriplex undulata. |
| PubMed: | A new method for the determination of carbonyl compounds in wines by headspace solid-phase microextraction coupled to gas chromatography-ion trap mass spectrometry. |
| PubMed: | Volatile compounds and sensory attributes of wine from Cv. Merlot (Vitis vinifera L.) grown under differential levels of water deficit with or without a kaolin-based, foliar reflectant particle film. |
| PubMed: | Volatile composition screening of Salix spp. nectar honey: benzenecarboxylic acids, norisoprenoids, terpenes, and others. |
| PubMed: | Contribution of microwave accelerated distillation in the extraction of the essential oil of Zygophyllum album L. |
| PubMed: | Analysis of carotenoids in grapes to predict norisoprenoid varietal aroma of wines from Apulia. |
| PubMed: | Identification and quantification of impact aroma compounds in 4 nonfloral Vitis vinifera varieties grapes. |
| PubMed: | Volatile compounds and the changes in their concentration levels during storage in beers containing varying malt concentrations. |
| PubMed: | Timing of cluster light environment manipulation during grape development affects C13 norisoprenoid and carotenoid concentrations in Riesling. |
| PubMed: | Carotenoid profiling and the expression of carotenoid biosynthetic genes in developing coffee grain. |
| PubMed: | Selectable one-dimensional or two-dimensional gas chromatography-mass spectrometry with simultaneous olfactometry or element-specific detection. |
| PubMed: | Influence of growth phase and geographic origin on the essential oil composition of Pituranthos chloranthus from Tunisia. |
| PubMed: | Characterization of the key aroma compounds in beef and pork vegetable gravies á la chef by application of the aroma extract dilution analysis. |
| PubMed: | Fate of key odorants in Sauternes wines through aging. |
| PubMed: | Identification of 3-hydroxy-beta-damascone and related carotenoid-derived aroma compounds as novel potent inducers of Nrf2-mediated phase 2 response with concomitant anti-inflammatory activity. |
| PubMed: | Volatile composition of Merlot wine from different vine water status. |
| PubMed: | Fate of grape flavor precursors during storage on yeast lees. |
| PubMed: | Carotenoid breakdown products the-norisoprenoids-in wine aroma. |
| PubMed: | Substrate promiscuity of RdCCD1, a carotenoid cleavage oxygenase from Rosa damascena. |
| PubMed: | Headspace solid-phase microextraction-gas chromatography-mass spectrometry for profiling free volatile compounds in Cabernet Sauvignon grapes and wines. |
| PubMed: | Effects of elevated CO2 on grapevine (Vitis vinifera L.): volatile composition, phenolic content, and in vitro antioxidant activity of red wine. |
| PubMed: | Instrumental and sensory approaches for the characterization of compounds responsible for wine aroma. |
| PubMed: | Characterization of the key aroma compounds in an american bourbon whisky by quantitative measurements, aroma recombination, and omission studies. |
| PubMed: | Free and hydrolytically released volatile compounds of Vitis vinifera L. cv. Fiano grapes as odour-active constituents of Fiano wine. |
| PubMed: | Characterization of the most odor-active compounds in an American Bourbon whisky by application of the aroma extract dilution analysis. |
| PubMed: | Identification of volatile compounds responsible for prune aroma in prematurely aged red wines. |
| PubMed: | The requisite level of theory for the computational design of molecularly imprinted silica xerogels. |
| PubMed: | The chemical characterization of the aroma of dessert and sparkling white wines (Pedro Ximénez, Fino, Sauternes, and Cava) by gas chromatography-olfactometry and chemical quantitative analysis. |
| PubMed: | Fragrance material review on cis-beta-damascone. |
| PubMed: | Fragrance material review on trans-beta-damascone. |
| PubMed: | Beta-damascenone, a suitable heat load indicator during wort boiling? |
| PubMed: | Comparative study of aromatic compounds in young red wines from cabernet sauvignon, cabernet franc, and cabernet gernischet varieties in China. |
| PubMed: | Release and formation of varietal aroma compounds during alcoholic fermentation from nonfloral grape odorless flavor precursors fractions. |
| PubMed: | Changes in key odorants of raw coffee beans during storage under defined conditions. |
| PubMed: | Characterization of the key aroma compounds in apricots (Prunus armeniaca) by application of the molecular sensory science concept. |
| PubMed: | Influence of plant water status on the production of C13-norisoprenoid precursors in Vitis vinifera L. Cv. cabernet sauvignon grape berries. |
| PubMed: | Which impact for beta-damascenone on red wines aroma? |
| PubMed: | A survey of seasonal temperatures and vineyard altitude influences on 2-methoxy-3-isobutylpyrazine, C13-norisoprenoids, and the sensory profile of Brazilian Cabernet Sauvignon wines. |
| PubMed: | Instrumental and sensory characterization of heat-induced odorants in aseptically packaged soy milk. |
| PubMed: | Aroma components of acid-hydrolyzed vegetable protein made by partial hydrolysis of rice bran protein. |
| PubMed: | Comparison of three lychee cultivar odor profiles using gas chromatography-olfactometry and gas chromatography-sulfur detection. |
| PubMed: | Aroma extraction dilution analysis of Sauternes wines. Key role of polyfunctional thiols. |
| PubMed: | Volatile composition and contribution to the aroma of spanish honeydew honeys. Identification of a new chemical marker. |
| PubMed: | Comparison of aroma volatiles in commercial Merlot and Cabernet Sauvignon wines using gas chromatography-olfactometry and gas chromatography-mass spectrometry. |
| PubMed: | Potent odorants characterize the aroma quality of leaves and stalks in raw and boiled celery. |
| PubMed: | Characterization of cachaça and rum aroma. |
| PubMed: | Thermal oxidation of 9'-cis-neoxanthin in a model system containing peroxyacetic acid leads to the potent odorant beta-damascenone. |
| PubMed: | Characterization of (E,E,Z)-2,4,6-nonatrienal as a character impact aroma compound of oat flakes. |
| PubMed: | Identification based on quantitative measurements and aroma recombination of the character impact odorants in a Bavarian Pilsner-type beer. |
| PubMed: | Aroma composition of red wines by different extraction methods and Gas Chromatography-SIM/MASS spectrometry analysis. |
| PubMed: | Identification of potent odorants formed during the preparation of extruded potato snacks. |
| PubMed: | Precursors to damascenone: synthesis and hydrolysis of isomeric 3,9-dihydroxymegastigma-4,6,7-trienes. |
| PubMed: | Analysis of hop-derived terpenoids in beer and evaluation of their behavior using the stir bar-sorptive extraction method with GC-MS. |
| PubMed: | Concurrent phenomena contributing to the formation of the aroma of wine during aging in oak wood: an analytical study. |
| PubMed: | Impact of growing environment on chickasaw blackberry (Rubus L.) aroma evaluated by gas chromatography olfactometry dilution analysis. |
| PubMed: | Identification and aroma impact of norisoprenoids in orange juice. |
| PubMed: | Fate of damascenone in wine: the role of SO2. |
| PubMed: | Analysis of the volatile aroma constituents of parental and hybrid clones of pepino (Solanum muricatum). |
| PubMed: | Simultaneous determination of E-2-nonenal and beta-damascenone in beer by reversed-phase liquid chromatography with UV detection. |
| PubMed: | Flavoring components of raw monsooned arabica coffee and their changes during radiation processing. |
| PubMed: | Impact odorants of different young white wines from the Canary Islands. |
| PubMed: | Prediction of aged red wine aroma properties from aroma chemical composition. Partial least squares regression models. |
| PubMed: | Identification of (3S, 9R)- and (3S, 9S)-megastigma-6,7-dien-3,5,9-triol 9-O-beta-D-glucopyranosides as damascenone progenitors in the flowers of Rosa damascena Mill. |
| PubMed: | Gas chromatographic-olfactometric characterization of aroma compounds in two types of cashew apple nectar. |
| PubMed: | Differential effects of tomato (Lycopersicon esculentum mill) matrix on the volatility of important aroma compounds. |
| PubMed: | Determination of volatile compounds in Grenache wines in relation with different terroirs in the Rhone Valley. |
| PubMed: | How low pH can intensify beta-damascenone and dimethyl trisulfide production through beer aging. |
| PubMed: | Character impact odorants of the apple cultivars Elstar and Cox Orange. |
| PubMed: | Effect of 1-methylcyclopropene on volatile emission and aroma in cv. Anna apples. |
| PubMed: | Chemical characterization of the aroma of Grenache rosé wines: aroma extract dilution analysis, quantitative determination, and sensory reconstitution studies. |
| PubMed: | Investigation of the beta-damascenone level in fresh and aged commercial beers. |
| PubMed: | Sensory and chemical changes in tomato sauces during storage. |
| PubMed: | Identification and quantification of aroma-active components that contribute to the distinct malty flavor of buckwheat honey. |
| PubMed: | Aroma composition changes in early season grapefruit juice produced from thermal concentration. |
| PubMed: | Monoterpenic and norisoprenoidic glycoconjugates of Vitis vinifera L. cv. Melon B. as precursors of odorants in Muscadet wines. |
| PubMed: | Volatile flavour components of baked potato flesh. A comparison of eleven potato cultivars. |
| PubMed: | 2-Methyl-3-furanthiol and methional are possible off-flavors in stored orange juice: aroma-similarity, NIF/SNIF GC-O, and GC analyses. |
| PubMed: | Use of solid-liquid distribution coefficients to determine retention properties of Porapak-Q resins. Determination of optimal conditions to isolate alkyl-methoxypyrazines and beta-damascenone from wine. |
| PubMed: | Change in the flavor of black tea drink during heat processing. |
| PubMed: | Identification and quantification of impact odorants of aged red wines from Rioja. GC-olfactometry, quantitative GC-MS, and odor evaluation of HPLC fractions. |
| PubMed: | Characterization of Volatiles in Rambutan Fruit (Nephelium lappaceum L.). |
| PubMed: | Olfactory and quantitative analysis of aroma compounds in elder flower (Sambucus nigra L.) drink processed from five cultivars. |
| PubMed: | Acid-catalyzed hydrolysis of alcohols and their beta-D-glucopyranosides. |
| PubMed: | Potent odorants of raw Arabica coffee. Their changes during roasting. |
| PubMed: | Identification of impact odorants in Bordeaux red grape juice, in the commercial yeast used for its fermentation, and in the produced wine. |
| PubMed: | Identification of potent odorants in Japanese green tea (Sen-cha). |
| PubMed: | Identification of character impact odorants of different soybean lecithins. |
| PubMed: | Differences in key odorants of handmade juice of yellow-flesh peaches (Prunus persica L.) induced by the workup procedure. |
| PubMed: | Quantification and sensory studies of character impact odorants of different soybean lecithins. |
| PubMed: | C(13)-Norisoprenoid glucoconjugates from lulo (Solanum quitoense L.) leaves. |
| PubMed: | Similarities in the aroma chemistry of Gewürztraminer variety wines and lychee (Litchi chinesis sonn.) fruit. |
| PubMed: | Quantitative determination of free and hydrolytically liberated beta-damascenone in red grapes and wines using a stable isotope dilution assay. |
| PubMed: | Production of β-damascenone precursors in cell cultures of Vitis labruscana cv Concord grapes. |
| PubMed: | Antispasmodic activity of beta-damascenone and E-phytol isolated from Ipomoea pes-caprae. |
| PubMed: | Progress in synthesis of sensory important trace components of essential oils and natural flavours. |
|
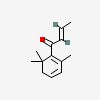 3D/inchi
3D/inchi



























