Articles:
lilial
Notes:
fresh accord - lilyall and benzyl isoeugenol.
autumn leaves accord - phenethyl phenyl acetate and lilyall and raspberry ketone.
clean fresh accord - aldehyde C-12 10% and lilyall.
cloth accord - musk gx, alcohol C-12, lilyall and santall.
| Fragrance Demo Formulas | ||
| CAS Number: | 80-54-6 | 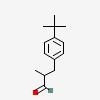 3D/inchi 3D/inchi
|
| Other(deleted CASRN): | 39390-70-0 | |
| ECHA EINECS - REACH Pre-Reg: | 201-289-8 | |
| FDA UNII: | T7540GJV69 | |
| Nikkaji Web: | J560.043J | |
| Beilstein Number: | 0880140 | |
| MDL: | MFCD00047655 | |
| XlogP3-AA: | 3.60 (est) | |
| Molecular Weight: | 204.31260000 | |
| Formula: | C14 H20 O | |
| BioActivity Summary: | listing | |
| NMR Predictor: | Predict (works with chrome, Edge or firefox) | |
Category: fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
| Google Scholar: | Search |
| Google Books: | Search |
| Google Scholar: with word "volatile" | Search |
| Google Scholar: with word "flavor" | Search |
| Google Scholar: with word "odor" | Search |
| Perfumer and Flavorist: | Search |
| Google Patents: | Search |
| US Patents: | Search |
| EU Patents: | Search |
| Pubchem Patents: | Search |
| PubMed: | Search |
| NCBI: | Search |
Physical Properties:
| Appearance: | colorless to pale yellow clear oily liquid (est) |
| Assay: | 93.00 to 100.00 % |
| Food Chemicals Codex Listed: | No |
| Specific Gravity: | 0.94200 to 0.94700 @ 25.00 °C. |
| Pounds per Gallon - (est).: | 7.838 to 7.880 |
| Refractive Index: | 1.50300 to 1.50600 @ 20.00 °C. |
| Boiling Point: | 250.00 to 251.00 °C. @ 750.00 mm Hg |
| Boiling Point: | 126.00 to 127.00 °C. @ 6.00 mm Hg |
| Acid Value: | 2.00 max. KOH/g |
| Vapor Pressure: | 0.005000 mmHg @ 25.00 °C. (est) |
| Flash Point: | 210.00 °F. TCC ( 98.89 °C. ) |
| logP (o/w): | 4.216 (est) |
| Shelf Life: | 24.00 month(s) or longer if stored properly. |
| Storage: | store in cool, dry place in tightly sealed containers, protected from heat and light. store under nitrogen. |
| Storage: | store under nitrogen. |
| Soluble in: | |
| alcohol | |
| paraffin oil | |
| water, 38 mg/L @ 20 °C (exp) | |
| Insoluble in: | |
| water | |
| Stability: | |
| detergent | |
| soap | |
Organoleptic Properties:
| Odor Type: floral | |
| Odor Strength: | medium |
| Substantivity: | 236 hour(s) at 100.00 % |
| floral muguet watery green powdery cumin | |
| Odor Description: at 100.00 %. | floral muguet watery green powdery cumin Luebke, William tgsc, (1986) |
| Odor and/or flavor descriptions from others (if found). | |
| Indukern F&F | |
| LILESTRALIS PURE | |
| Odor Description: | FRESH, GREEN, FLORAL, LILAC |
| Prodasynth | |
| LILIAL (> 99% | |
| Odor Description: | FRESH, LIGHT, FLORAL,MUGUET,POWDERY |
Cosmetic Information:
| CosIng: | cosmetic data |
| Cosmetic Uses: |
perfuming agents |
Suppliers:
Safety Information:
| Preferred SDS: View | |
| European information : | |
| Most important hazard(s): | |
| Xn - Harmful. | |
|
R 22 - Harmful if swallowed. R 36/38 - Irritating to skin and eyes. R 43 - May cause sensitisation by skin contact. S 02 - Keep out of the reach of children. S 24/25 - Avoid contact with skin and eyes. S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S 37 - Wear suitable gloves. | |
| Hazards identification | |
| Classification of the substance or mixture | |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) | |
| Acute toxicity, Oral (Category 4), H302 Skin irritation (Category 2), H315 Skin sensitisation (Category 1), H317 Reproductive toxicity (Category 2), H361 Acute aquatic toxicity (Category 2), H401 Chronic aquatic toxicity (Category 2), H411 | |
| GHS Label elements, including precautionary statements | |
| Pictogram |    |
| Signal word | Warning |
| Hazard statement(s) | |
| H302 - Harmful if swallowed H315 - Causes skin irritation H317 - May cause an allergic skin reaction H361 - Suspected of damaging fertility or the unborn child H401 - Toxic to aquatic life H411 - Toxic to aquatic life with long lasting effects | |
| Precautionary statement(s) | |
| P201 - Obtain special instructions before use. P202 - Do not handle until all safety precautions have been read and understood. P261 - Avoid breathing dust/fume/gas/mist/vapours/spray. P264 - Wash skin thouroughly after handling. P270 - Do not eat, drink or smoke when using this product. P272 - Contaminated work clothing should not be allowed out of the workplace. P273 - Avoid release to the environment. P280 - Wear protective gloves/protective clothing/eye protection/face protection. P281 - Use personal protective equipment as required. P301 + P312 - IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302 + P352 - IF ON SKIN: wash with plenty of soap and water. P308 + P313 - IF exposed or concerned: Get medical advice/attention. P330 - Rinse mouth. P333 + P313 - IF SKIN irritation or rash occurs: Get medical advice/attention. P362 - Take off contaminated clothing and wash before reuse. P391 - Collect spillage. Hazardous to the aquatic environment P405 - Store locked up. P501 - Dispose of contents/container in accordance with local/regional/national/international regulations. | |
| Human Experience: | |
| 4 % solution: no irritation or sensitization. | |
| Oral/Parenteral Toxicity: | |
|
oral-rat LD50 1390 mg/kg BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: DYSPNEA National Technical Information Service. Vol. OTS0535449 intraperitoneal-mouse LD50 700 mg/kg National Technical Information Service. Vol. OTS0535449 | |
| Dermal Toxicity: | |
|
skin-rabbit LD50 > 5000 mg/kg National Technical Information Service. Vol. OTS0535449 | |
| Inhalation Toxicity: | |
| Not determined | |
Safety in Use Information:
| Category: | fragrance agents | ||
| RIFM Fragrance Material Safety Assessment: Search | |||
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice | |||
| IFRA Critical Effect: | Dermal sensitization and systemic toxicity | ||
| contains the following IFRA (Annex) restricted components: (non-analysis max. level reference only) | |||
| lilyall / methyl anthranilate schiff's base | Max. Found: 60.60 % and Reason: Sensitization | ||
| IFRA: | View Standard | ||
| View IFRA Standards Library for complete information. | |||
| Please review Amendment 49 IFRA documentation for complete information. | |||
| IFRA RESTRICTION LIMITS IN THE FINISHED PRODUCT (%): | |||
| Category 1: Products applied to the lips | |||
| Prohibited % | |||
| Category 2: Products applied to the axillae | |||
| 0.09 % | |||
| Category 3: Products applied to the face/body using fingertips | |||
| 0.04 % | |||
| Category 4: Products related to fine fragrance | |||
| 1.40 % | |||
| Category 5: Products applied to the face and body using the hands (palms), primarily leave-on | |||
| Category 5A: Body lotion products applied to the body using the hands (palms), primarily leave-on | |||
| 0.06 % | |||
| Category 5B: Face moisturizer products applied to the face using the hands (palms), primarily leave-on | |||
| 0.05 % | |||
| Category 5C: Hand cream products applied to the hands using the hands (palms), primarily leave-on | |||
| 0.05 % | |||
| Category 5D: Baby Creams, baby Oils and baby talc | |||
| 0.017 % | |||
| Category 6: Products with oral and lip exposure | |||
| Prohibited % | |||
| Category 7: Products applied to the hair with some hand contact | |||
| Category 7A: Rinse-off products applied to the hair with some hand contact | |||
| 0.04 % | |||
| Category 7B: Leave-on products applied to the hair with some hand contact | |||
| 0.04 % | |||
| Category 8: Products with significant anogenital exposure | |||
| 0.017 % | |||
| Category 9: Products with body and hand exposure, primarily rinse off | |||
| 0.10 % | |||
| Category 10: Household care products with mostly hand contact | |||
| Category 10A: Household care excluding aerosol products (excluding aerosol/spray products) | |||
| 0.10 % | |||
| Category 10B: Household aerosol/spray products | |||
| 0.63 % | |||
| Category 11: Products with intended skin contact but minimal transfer of fragrance to skin from inert substrate | |||
| Category 11A: Products with intended skin contact but minimal transfer of fragrance to skin from inert substrate without UV exposure | |||
| 0.017 % | |||
| Category 11B: Products with intended skin contact but minimal transfer of fragrance to skin from inert substrate with potential UV exposure | |||
| 0.017 % | |||
| Category 12: Products not intended for direct skin contact, minimal or insignificant transfer to skin | |||
| 16.00 % | |||
| Notes: | |||
| IFRA FLAVOR REQUIREMENTS: | |||
Due to the possible ingestion of small amounts of fragrance ingredients from their use in products in Categories 1 and 6, materials must not only comply with IFRA Standards but must also be recognized as safe as a flavoring ingredient as defined by the IOFI Code of Practice (www.iofi.org). For more details see chapter 1 of the Guidance for the use of IFRA Standards. | |||
| Recommendation for lilyall flavor usage levels up to: | |||
| not for flavor use. | |||
Safety References:
| EPI System: | View |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA Substance Registry Services (TSCA): | 80-54-6 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 6652 |
| National Institute of Allergy and Infectious Diseases: | Data |
| SCCNFP: | opinion |
| WISER: | UN 3082 |
| WGK Germany: | 3 |
| 3-(4-tert-butylphenyl)butanal | |
| Chemidplus: | 0000080546 |
| RTECS: | MW4895000 for cas# 80-54-6 |
References:
| Leffingwell: | Chirality or Article |
| 3-(4-tert-butylphenyl)butanal | |
| NIST Chemistry WebBook: | Search Inchi |
| Canada Domestic Sub. List: | 80-54-6 |
| Pubchem (cid): | 6652 |
| Pubchem (sid): | 134972321 |
Other Information:
| (IUPAC): | Atomic Weights of the Elements 2011 (pdf) |
| Videos: | The Periodic Table of Videos |
| tgsc: | Atomic Weights use for this web site |
| (IUPAC): | Periodic Table of the Elements |
| HMDB (The Human Metabolome Database): | Search |
| Export Tariff Code: | 2912.49.2600 |
| ChemSpider: | View |
| Wikipedia: | View |
Potential Blenders and core components note
Potential Uses:
| alliage | ||
| anais anais | ||
| azzaro | ||
| blue mist | FR | |
| bouquet | FR | |
| bubble gum | FR | |
| calyx | ||
| cedar | FR | |
| charlie | ||
| cherry blossom | FR | |
| citrus | FR | |
| clean | ||
| clean linen | FR | |
| coconut tropical coconut | FR | |
| country meadow | FR | |
| cyclamen | FR | |
| devin | ||
| estee | ||
| eternity | ||
| fern | FR | |
| floral | FR | |
| gardenia | FR | |
| grapefruit | FR | |
| green | FR | |
| halston | ||
| herbal | FR | |
| honeysuckle | FR | |
| j' ai ose | ||
| lilac | FR | |
| lily | FR | |
| lily of the valley | FR | |
| linden flower | FR | |
| lotus | FR | |
| marbert man | ||
| mint | FR | |
| musk | FR | |
| new car | FR | |
| paco rabanne | ||
| pine | FR | |
| potpourri | FR | |
| pour homme | ||
| rain | FR | |
| rose | FR | |
| sandalwood | FR | |
| sea breeze | FR | |
| spring rain | FR | |
| tide | ||
| turbulences | ||
| woody | FR |
Occurrence (nature, food, other): note
| not found in nature |
Synonyms:
| aldehyde MBDC | |
| aldehyde MBDC pure | |
| 2-(4-tert- | butyl benzyl) propionaldehyde |
| butyl phenyl methyl propional | |
| p-tert- | butyl-a-methyldihydrocinnamic aldehyde |
| p-tert- | butyl-a-methylhydrocinnamaldehyde |
| p-tert- | butyl-a-methylhydrocinnamic aldehyde |
| p-tert- | butyl-alpha-methyl dihydrocinnamaldehyde |
| para-tert- | butyl-alpha-methyl dihydrocinnamaldehyde |
| meta-tert- | butyl-alpha-methyl dihydrocinnamic aldehyde |
| p-tert- | butyl-alpha-methyl dihydrocinnamic aldehyde |
| para-tert- | butyl-alpha-methyl dihydrocinnamic aldehyde |
| p-tert- | butyl-alpha-methyl hydrocinnamaldehyde |
| para-tert- | butyl-alpha-methyl hydrocinnamaldehyde |
| p-tert- | butyl-alpha-methyl hydrocinnamic aldehyde |
| para-tert- | butyl-alpha-methyl hydrocinnamic aldehyde |
| m-tert- | butyl-alpha-methyldihydrocinnamic aldehyde |
| 2-(4-tert- | butylbenzyl)propionaldehyde |
| 3-(4-tert- | butylphenyl)butanal |
| butylphenylmethylpropional | |
| 4-(1,1- | dimethyl ethyl)-alpha-methyl benzene propanal |
| 4-(1,1- | dimethylethyl)-alpha-methylbenzenepropanal |
| hydrocinnamaldehyde, p-tert-butyl-a-methyl- | |
| lilestralis | |
| lilestralis pure | |
| lilial | |
| lilialdehyde | |
| liligul N 743 CLP | |
| lilyall | |
| lysmeral | |
| lysmeral extra | |
| mefloral | |
| 3-[4-(2- | methyl-2-propanyl)phenyl]butanal |
| alpha- | methyl-beta-(p-tert-butylphenyl)propionaldehyde |
| alpha- | methyl-beta-(para-tert-butyl phenyl) propionaldehyde |
| a- | methyl-p-(tert-butyl)hydrocinnamaldehyde |
| alpha- | methyl-p-(tert-butyl)hydrocinnamaldehyde |
| alpha- | methyl-para-(tert-butyl) hydrocinnamaldehyde |




















