|
Category: flavor and fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
Physical Properties:
| Appearance: | white crystalline solid (est) |
| Assay: | 98.00 to 100.00 %
|
| Food Chemicals Codex Listed: | No |
| Melting Point: | 56.00 to 60.00 °C. @ 760.00 mm Hg
|
| Boiling Point: | 210.00 to 213.00 °C. @ 760.00 mm Hg
|
| Boiling Point: | 109.00 °C. @ 20.00 mm Hg
|
| Vapor Pressure: | 0.250000 mmHg @ 25.00 °C. (est) |
| Vapor Density: | 4.8 ( Air = 1 ) |
| Flash Point: | > 212.00 °F. TCC ( > 100.00 °C. )
|
| logP (o/w): | 2.040 |
| Shelf Life: | 36.00 month(s) or longer if stored properly. |
| Storage: | store in cool, dry place in tightly sealed containers, protected from heat and light. |
| Soluble in: |
| | alcohol | | | water, 1543 mg/L @ 25 °C (est) |
| Insoluble in: |
| | water |
| Stability: |
| | non-discoloring in most media |
Organoleptic Properties:
| |
| Odor Type: green |
| |
| Odor Strength: | high ,
recommend smelling in a 1.00 % solution or less |
| |
| Substantivity: | 76 hour(s) at 100.00 % |
| |
| | sweet green hay new mown hay fennel |
Odor Description:
at 1.00 % in dipropylene glycol. | sweet green new mown hay fennel
Luebke, William tgsc, (1981) |
| |
| |
| Flavor Type: green |
| |
| | green hawthorn hay new mown hay anisic anise fennel powdery fatty |
Taste Description:
| sweet green hawthorn new mown hay anisic anise fennel powdery fatty
Luebke, William tgsc, (1981) |
| |
| Odor and/or flavor descriptions from others (if found). |
| |
| Moellhausen |
| DIMETHYL HYDROQUINONE |
| Odor Description: | sweet, aromatic, erbaceous, earthy, tobacco note |
| Taste Description: | aromatic, dusty, earthy |
| |
| |
Cosmetic Information:
Suppliers:
| Augustus Oils |
| Dimethyl Hydro Quinone
|
| Services |
| Beijing Lys Chemicals |
| 1,4-Dimethoxy benzene
|
| Berjé |
| Dimethyl Hydroquinone
|
| Media |
| BOC Sciences |
| For experimental / research use only. |
| para-Dimethyl Hydroquinone
|
| Creatingperfume.com |
| Dimethyl Hydroquinone (1,4-Dimethoxybenzene)
Odor: Sweet, green, new mown hay, fennel, aromatic, herbaceous, earthy, tobacco note |
| Diffusions Aromatiques |
| 1.4-DIMETHOXYBENZENE
|
| EMD Millipore |
| For experimental / research use only. |
| Hydroquinone dimethyl ether
|
| Ernesto Ventós |
| DIMETHYL HYDROQUINONE
|
| Indukern F&F |
| DIMETHYL HYDROQUINONE
Odor: WARM, HERBAL, POWERFUL, SWEET |
| Keva |
| DIMETHYL HYDROQUINONE
Odor: Warm, herbaceous sweet |
| Lluch Essence |
| DIMETHYL HYDROQUINONE
|
| M&U International |
| 1,4-DIMETHOXYBENZENE, Kosher
|
| Moellhausen |
| DIMETHYL HYDROQUINONE
Odor: sweet, aromatic, erbaceous, earthy, tobacco note Flavor: aromatic, dusty, earthy |
| Pell Wall Perfumes |
| Dimethyl Hydroquinone
|
| Penta International |
| DIMETHYL HYDROQUINONE
|
| R C Treatt & Co Ltd |
| Dimethyl Hydroquinone
|
| Santa Cruz Biotechnology |
| For experimental / research use only. |
| 1.4-Dimethoxybenzene 98%
|
| Sigma-Aldrich |
| 1,4-Dimethoxybenzene, 99%, FG
Odor: fennel; fatty. |
| Certified Food Grade Products |
| SRS Aromatics |
| DIMETHYL HYDROQUINONE
|
| Synerzine |
| p-Dimethoxybenzene
|
| TCI AMERICA |
| For experimental / research use only. |
| 1.4-Dimethoxybenzene >99.0%(GC)
|
| Tianjin Danjun International |
| 1,4-Dimethoxy benzene
|
| WholeChem |
| 1,4-Dimethoxy benzene
|
Safety Information:
| Preferred SDS: View |
| European information : |
| Most important hazard(s): | | Xi - Irritant |
R 36/38 - Irritating to skin and eyes.
S 02 - Keep out of the reach of children.
S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S 36 - Wear suitable protective clothing.
|
| |
| Hazards identification |
| |
| Classification of the substance or mixture |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) |
Skin irritation (Category 2), H315
Eye irritation (Category 2A), H319
Specific target organ toxicity - single exposure (Category 3), Respiratory system, H335
|
| GHS Label elements, including precautionary statements |
| |
| Pictogram |  |
| |
| Signal word | Warning |
| Hazard statement(s) |
H315 - Causes skin irritation
H319 - Causes serious eye irritation
H335 - May cause respiratory irritation
|
| Precautionary statement(s) |
P261 - Avoid breathing dust/fume/gas/mist/vapours/spray.
P264 - Wash skin thouroughly after handling.
P271 - Use only outdoors or in a well-ventilated area.
P280 - Wear protective gloves/protective clothing/eye protection/face protection.
P302 + P352 - IF ON SKIN: wash with plenty of soap and water.
P304 + P340 - IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing.
P305 + P351 + P338 - IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
P312 - Call a POISON CENTER or doctor/physician if you feel unwell.
P332 + P313 - IF SKIN irritation occurs: Get medical advice/attention.
P337 + P313 - IF eye irritation persists: Get medical advice/attention.
P362 - Take off contaminated clothing and wash before reuse.
P403 + P233 - Store in a well-ventilated place. Keep container tightly closed.
P405 - Store locked up.
P501 - Dispose of contents/ container to an approved waste disposal plant.
|
| Human Experience: |
| 4 % solution: no irritation or sensitization. |
| Oral/Parenteral Toxicity: |
oral-rat LD50 3600 mg/kg
(Moreno, 1973ae)
oral-rat LD50 8500 mg/kg
(Moreno, 1973ae)
oral-mouse LD50 4000 mg/kg
BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY)
BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY)
Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 4(2), Pg. 30, 1960.
intraperitoneal-mouse LD50 100 mg/kg
National Technical Information Service. Vol. AD277-689
oral-rat LD50 3600 mg/kg
Food and Cosmetics Toxicology. Vol. 16, Pg. 715, 1978.
|
| Dermal Toxicity: |
skin-rabbit LD50 > 5000 mg/kg
Food and Cosmetics Toxicology. Vol. 16, Pg. 715, 1978.
|
| Inhalation Toxicity: |
inhalation-rat LC50 500 mg/24hours
Food and Cosmetics Toxicology. Vol. 16, Pg. 715, 1978.
|
Safety in Use Information:
| Category: | flavor and fragrance agents |
| RIFM Fragrance Material Safety Assessment: Search |
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice |
| Recommendation for para-dimethyl hydroquinone usage levels up to: | | | 2.0000 % in the fragrance concentrate.
|
| |
| Maximised Survey-derived Daily Intakes (MSDI-EU): | 15.00 (μg/capita/day) |
| Maximised Survey-derived Daily Intakes (MSDI-USA): | 7.00 (μg/capita/day) |
| Structure Class: | I |
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). |
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library |
| publication number: 3 |
| Click here to view publication 3 |
| | average usual ppm | average maximum ppm |
| baked goods: | - | 5.80000 |
| beverages(nonalcoholic): | - | 8.10000 |
| beverages(alcoholic): | - | - |
| breakfast cereal: | - | - |
| cheese: | - | - |
| chewing gum: | - | - |
| condiments / relishes: | - | - |
| confectionery froastings: | - | - |
| egg products: | - | - |
| fats / oils: | - | - |
| fish products: | - | - |
| frozen dairy: | - | 5.00000 |
| fruit ices: | - | 5.00000 |
| gelatins / puddings: | - | - |
| granulated sugar: | - | - |
| gravies: | - | - |
| hard candy: | - | 4.70000 |
| imitation dairy: | - | - |
| instant coffee / tea: | - | - |
| jams / jellies: | - | - |
| meat products: | - | - |
| milk products: | - | - |
| nut products: | - | - |
| other grains: | - | - |
| poultry: | - | - |
| processed fruits: | - | - |
| processed vegetables: | - | - |
| reconstituted vegetables: | - | - |
| seasonings / flavors: | - | - |
| snack foods: | - | - |
| soft candy: | - | - |
| soups: | - | - |
| sugar substitutes: | - | - |
| sweet sauces: | - | - |
Safety References:
| European Food Safety Athority(EFSA): | Flavor usage levels; Subacute, Subchronic, Chronic and Carcinogenicity Studies; Developmental / Reproductive Toxicity Studies; Genotoxicity Studies... |
| European Food Safety Authority (EFSA) reference(s): |
Opinion of the Scientific Panel on food additives, flavourings, processing aids and materials in contact with food (AFC) related to Flavouring Group Evaluation 23 (FGE.23): Aliphatic, alicyclic and aromatic ethers including anisole derivatives From chemical groups 15, 16 and 26 (Commission Regulation (EC) No 1565/2000 of 18 July 2000
View page or View pdf |
Flavouring Group Evaluation 59 (FGE.59): Consideration of aliphatic and aromatic ethers evaluated by JECFA (61st meeting) structurally related to aliphatic, alicyclic and aromatic ethers including anisole derivatives evaluated by EFSA in FGE.23 (2006) (Commission Regulation (EC) No 1565/2000 of 18 July 2000) - Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in contact with Food (AFC)
View page or View pdf |
Flavouring Group Evaluation 23, Revision 1 (FGE.23Rev1): Aliphatic, alicyclic and aromatic ethers including anisole derivatives from chemical groups 15, 16, 26 and 30[1] - Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food
View page or View pdf |
Scientific Opinion on Flavouring Group Evaluation 59, Revision 1 (FGE.59Rev1): Consideration of aliphatic and aromatic ethers evaluated by JECFA (61st meeting and 63rd meeting) structurally related to aliphatic, alicyclic and aromatic ethers including anisole derivatives evaluated by EFSA in FGE.23 Rev2 (2010)
View page or View pdf |
Scientific Opinion on the safety and efficacy of aromatic ethers including anisole derivatives (chemical group 26) when used as feed additives for all animal species
View page or View pdf |
| EPI System: | View |
| NIOSH International Chemical Safety Cards: | search |
| Chemical Carcinogenesis Research Information System: | Search |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA Substance Registry Services (TSCA): | 150-78-7 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 9016 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WGK Germany: | 1 |
| | 1,4-dimethoxybenzene |
| Chemidplus: | 0000150787 |
| EPA/NOAA CAMEO: | hazardous materials |
| RTECS: | 150-78-7 |
References:
Other Information:
Potential Blenders and core components note
| |
| For Odor |
| No odor group found for these |
| 2- | acetonyl-5-methyl furan | |
| 1- | amyl-pyrrole-3-aldehyde | |
| 2,5- | dimethyl-3,6-dihexyl pyrazine | |
| 2-( | pyridyl-2)-ethyl ethyl sulfide | |
| para- | thiocresol | |
| aldehydic |
| | muguet undecadienal | FR |
| 2- | tridecenal | FL/FR |
| amber |
| | cistus ladaniferus resinoid | FL/FR |
| animal |
| iso | butyl quinoline | FR |
| iso | butyl quinoline | FR |
| para- | cresyl caprylate | FL/FR |
| para- | cresyl phenyl acetate | FL/FR |
| | indole | FL/FR |
| anisic |
| | ocimum basilicum herb oil | FL/FR |
| balsamic |
| iso | amyl benzoate | FL/FR |
| | amyris wood oil | FL/FR |
| | benzophenone | FR |
| | benzyl benzoate | FL/FR |
| | benzyl salicylate | FL/FR |
| | cinnamyl alcohol | FL/FR |
| | cinnamyl formate | FL/FR |
| | clover nitrile | FR |
| | ethyl cinnamate | FL/FR |
| | fir balsam absolute | FR |
| | geranyl benzoate | FL/FR |
| | methyl cinnamate | FL/FR |
| 2- | phenyl propyl alcohol | FL/FR |
| 3- | phenyl propyl alcohol | FL/FR |
| | prenyl benzoate | FL/FR |
| | terpinyl benzoate | FR |
| brown |
| sec- | heptyl acetate | FL/FR |
| chemical |
| | styralyl alcohol | FL/FR |
| citrus |
| | citral dimethyl acetal | FL/FR |
| (E)-4- | decenal | FL/FR |
| | dihydromyrcenol | FL/FR |
| | grapefruit pentanol | FR |
| coumarinic |
| | coumane | FL/FR |
| ethereal |
| | acetaldehyde dimethyl acetal | FL/FR |
| iso | valeraldehyde propylene glycol acetal | FL/FR |
| fatty |
| | decanol | FL/FR |
| 2- | decenal | FL/FR |
| 2- | nonen-1-ol | FL/FR |
| 2- | nonenal | FL/FR |
| fermented |
| | hexanal diethyl acetal | FL/FR |
| floral |
| | amyl benzoate | FL/FR |
| alpha- | amyl cinnamaldehyde | FL/FR |
| iso | amyl salicylate | FL/FR |
| | anisyl propanal / methyl anthranilate schiff's base | FR |
| alpha- | butyl cinnamaldehyde | FL/FR |
| iso | butyl salicylate | FL/FR |
| | champaca absolute | FR |
| | citronellol | FL/FR |
| | citronellyl acetate | FL/FR |
| | citronellyl propionate | FL/FR |
| | coriander seed oil | FL/FR |
| | cumin carbinol | FR |
| | cuminyl acetaldehyde | FL/FR |
| | dimethyl benzyl carbinol | FL/FR |
| | dimethyl benzyl carbinyl butyrate | FL/FR |
| 6,8- | dimethyl-2-nonanol | FR |
| | ethyl 2-benzyl butyrate | FL/FR |
| | ethyl hydrocinnamate | FL/FR |
| | ethyl ortho-anisate | FL/FR |
| | farnesyl acetate | FL/FR |
| | floral butanal | FR |
| | floral pyranol | FR |
| | gardenia absolute | FR |
| | geranium oil bourbon | FL/FR |
| | geranyl acetate | FL/FR |
| (E)- | geranyl acetone | FL/FR |
| | geranyl acetone | FL/FR |
| | geranyl isobutyrate | FL/FR |
| | geranyl phenyl acetate | FL/FR |
| | heliotropin | FL/FR |
| (Z)-3- | hexen-1-yl salicylate | FL/FR |
| alpha- | hexyl cinnamaldehyde | FL/FR |
| | hyacinth ether | FR |
| | hydroxycitronellal diethyl acetal | FL/FR |
| | jasmin cyclopentanol | FR |
| | jonquil absolute | FR |
| | kewda absolute | |
| | kewda oil | CS |
| laevo- | linalool | FL/FR |
| | methyl dihydrojasmonate | FL/FR |
| | mimosa absolute france | FL/FR |
| | neroli oil bigarde | FL/FR |
| | nerolidol | FL/FR |
| | ocean propanal | FL/FR |
| | ocean propanal / methyl anthranilate schiff's base | FR |
| | orange leaf absolute | FL/FR |
| bitter | orangeflower absolute morocco | FL/FR |
| | papaya isobutyrate | FL/FR |
| | phenethyl alcohol | FL/FR |
| | phenethyl formate | FL/FR |
| | phenethyl salicylate | FL/FR |
| | phenyl acetaldehyde / methyl anthranilate schiff's base | FR |
| | phenyl acetaldehyde diisobutyl acetal | FL/FR |
| | phenyl glycol diacetate | FR |
| 2- | phenyl propionaldehyde dimethyl acetal | FL/FR |
| 2- | phenyl propionaldehyde ethylene glycol acetal | FR |
| 2- | phenyl propyl acetate | FL/FR |
| (R)-2- | phenyl propyl alcohol | FL/FR |
| 3- | phenyl propyl formate | FL/FR |
| | phenyl propyl phenyl acetate | FR |
| 3- | phenyl propyl propionate | FL/FR |
| (E)-2- | phenyl-1(2)-propene-1-yl acetate | FR |
| | reseda acetal | FR |
| | rose butanoate | FL/FR |
| | styralyl formate | FL/FR |
| | sweet pea absolute | FR |
| | tea acetate | FR |
| | terpinyl isobutyrate | FL/FR |
| | tetrahydrolinalool | FL/FR |
| | tuberose absolute (from concrete) | FL/FR |
| | tuberose absolute (from pommade) | FL/FR |
| | tuberose acetate | FR |
| | verdyl acetate | FR |
| | violet methyl carbonate | FR |
| fruity |
| | allyl amyl glycolate | FR |
| | allyl cyclohexyl propionate | FL/FR |
| iso | amyl butyrate | FL/FR |
| iso | amyl isobutyrate | FL/FR |
| iso | amyl isovalerate | FL/FR |
| | benzyl methyl ether | FL/FR |
| | diethyl succinate | FL/FR |
| | dimethyl succinate | FL/FR |
| | ethyl methyl-para-tolyl glycidate | FL/FR |
| | green acetate | FR |
| 2- | hexen-1-ol | FL/FR |
| | hexyl acetate | FL/FR |
| | peach pivalate | FR |
| green |
| | acetaldehyde ethyl phenethyl acetal | FL/FR |
| iso | amyl 3-(2-furan) propionate | FL/FR |
| iso | butyl benzyl carbinol | FL/FR |
| | chrysanthemum oxide | FL/FR |
| | citrus carbaldehyde / methyl anthranilate schiff's base | FR |
| | decanal propylene glycol acetal | FL/FR |
| | diphenyl methane | FL/FR |
| | diphenyl oxide | FL/FR |
| | earthy acetal | FL/FR |
| | galbascone (IFF) | FR |
| | green ether | FL/FR |
| 1- | heptanol | FL/FR |
| | heptyl acetate | FL/FR |
| | heptyl cinnamate | FL/FR |
| (Z)-3- | hexen-1-yl benzoate | FL/FR |
| | hexyl tiglate | FL/FR |
| | hyacinth absolute | FL/FR |
| | hyacinth butanal | FR |
| | ivy carbaldehyde / methyl anthranilate schiff's base | FR |
| | ivy dioxolane | FR |
| (Z)- | leaf acetal | FL/FR |
| | leafy acetal | FL/FR |
| | melon nonenoate | FL/FR |
| | methyl cyclocitrone (IFF) | FR |
| para- | methyl hydratropaldehyde | FL/FR |
| | methyl octine carbonate | FL/FR |
| 4- | methyl-4-phenyl pentanone | FR |
| | narcissus flower absolute | FR |
| (Z)-2- | nonen-1-ol | FL/FR |
| (E)-2- | nonen-1-ol | FL/FR |
| | octanal dimethyl acetal | FL/FR |
| (E)-2- | octen-1-yl acetate | FL/FR |
| | octyl oxyacetaldehyde | FR |
| | olive oil absolute | FL/FR |
| | phenethyl oxyacetaldehyde | FR |
| | phenoxyacetaldehyde 50% in benzyl alcohol | FR |
| | phenyl acetaldehyde | FL/FR |
| | phenyl acetaldehyde dimethyl acetal | FL/FR |
| | phenyl acetaldehyde ethylene glycol acetal | FR |
| 3- | phenyl propionaldehyde | FL/FR |
| 2- | phenyl propionaldehyde | FL/FR |
| | styralyl acetate | FL/FR |
| | violet leaf absolute | FL/FR |
| herbal |
| | clary sage absolute | FL/FR |
| | clary sage oil france | FL/FR |
| | daucus carota fruit oil | FL/FR |
| | lavender absolute bulgaria | FL/FR |
| | methyl ortho-anisate | FL/FR |
| beta- | pinene | FL/FR |
| laevo-beta- | pinene | FL/FR |
| honey |
| | phenyl acetic acid | FL/FR |
| marine |
| green | algae absolute | FL/FR |
| | marine hexane | FR |
| melon |
| (Z)-6- | nonen-1-yl acetate | FL/FR |
| minty |
| | spearmint absolute | FL/FR |
| 2,4,4,6- | tetramethyl cyclohexa-2,5-diene-1-one | FR |
| mossy |
| | oakmoss absolute | FL/FR |
| | veramoss (IFF) | FR |
| naphthyl |
| para- | methyl anisole | FL/FR |
| powdery |
| para- | anisyl alcohol | FL/FR |
| spicy |
| | clove bud oil | FL/FR |
| | cuminaldehyde | FL/FR |
| black | currant bud absolute | FL/FR |
| | eugenol | FL/FR |
| | pimenta acris leaf oil | FL/FR |
| terpenic |
| | cypress leaf oil | FR |
| D-(+)-beta- | pinene | FL/FR |
| alpha- | terpineol | FL/FR |
| tonka |
| | coumarin | FR |
| | tonka bean absolute | FR |
| vanilla |
| | ethyl vanillin | FL/FR |
| | vanilla bean absolute (vanilla planifolia) | FL/FR |
| | vanillyl acetate | FL/FR |
| waxy |
| 3- | decanone | FL/FR |
| | ethyl laurate | FL/FR |
| woody |
| | guaiacwood oil | FL/FR |
| | santall | FR |
| | vetiver oil haiti | FL/FR |
| | woody acetate | FR |
| (Z)- | woody amylene | FR |
| |
| For Flavor |
| |
| No flavor group found for these |
| green | algae absolute | FL/FR |
| | amyl benzoate | FL/FR |
| | cistus ladaniferus resinoid | FL/FR |
| | coumane | FL/FR |
| | decanal propylene glycol acetal | FL/FR |
| 3- | decanone | FL/FR |
| | earthy acetal | FL/FR |
| | ethyl 2-benzyl butyrate | FL/FR |
| | ethyl hydrocinnamate | FL/FR |
| | ethyl ortho-anisate | FL/FR |
| | geranyl benzoate | FL/FR |
| | heptyl cinnamate | FL/FR |
| | hexanal diethyl acetal | FL/FR |
| | hyacinth absolute | FL/FR |
| | kewda absolute | |
| | methyl ortho-anisate | FL/FR |
| 2- | phenyl propyl acetate | FL/FR |
| (R)-2- | phenyl propyl alcohol | FL/FR |
| 3- | phenyl propyl formate | FL/FR |
| 3- | phenyl propyl propionate | FL/FR |
| laevo-beta- | pinene | FL/FR |
| D-(+)-beta- | pinene | FL/FR |
| | prenyl benzoate | FL/FR |
| | styralyl formate | FL/FR |
| | terpinyl isobutyrate | FL/FR |
|
| iso | amyl 3-(2-furan) propionate | FL/FR |
| alpha- | butyl cinnamaldehyde | FL/FR |
| aldehydic |
| 2- | tridecenal | FL/FR |
| amber |
| iso | butyl benzyl carbinol | FL/FR |
| animal |
| para- | cresyl caprylate | FL/FR |
| | indole | FL/FR |
| aromatic |
| | leafy acetal | FL/FR |
| astringent |
| 2-( | pyridyl-2)-ethyl ethyl sulfide | |
| balsamic |
| | benzyl benzoate | FL/FR |
| | benzyl salicylate | FL/FR |
| | ethyl cinnamate | FL/FR |
| burnt |
| para- | thiocresol | |
| chemical |
| | styralyl alcohol | FL/FR |
| cherry |
| | heliotropin | FL/FR |
| citrus |
| | citral dimethyl acetal | FL/FR |
| laevo- | linalool | FL/FR |
| alpha- | terpineol | FL/FR |
| cooling |
| iso | butyl salicylate | FL/FR |
| cucumber |
| 2- | ethyl octine carbonate | FL |
| ethereal |
| | acetaldehyde dimethyl acetal | FL/FR |
| fatty |
| 2- | acetonyl-5-methyl furan | |
| 1- | amyl-pyrrole-3-aldehyde | |
| 2- | decenal | FL/FR |
| (E,E)-2,4- | heptadienal | FL |
| sec- | heptyl acetate | FL/FR |
| (Z)-3- | hexen-1-yl benzoate | FL/FR |
| 2- | nonen-1-ol | FL/FR |
| (Z)-2- | nonen-1-ol | FL/FR |
| 2- | nonenal | FL/FR |
| floral |
| | citronellol | FL/FR |
| | citronellyl acetate | FL/FR |
| | citronellyl propionate | FL/FR |
| | dimethyl benzyl carbinyl butyrate | FL/FR |
| | farnesyl acetate | FL/FR |
| | geranium oil bourbon | FL/FR |
| (E)- | geranyl acetone | FL/FR |
| | geranyl acetone | FL/FR |
| | geranyl isobutyrate | FL/FR |
| | geranyl phenyl acetate | FL/FR |
| | methyl dihydrojasmonate | FL/FR |
| | neroli oil bigarde | FL/FR |
| | ocean propanal | FL/FR |
| | orange leaf absolute | FL/FR |
| bitter | orangeflower absolute morocco | FL/FR |
| | phenethyl alcohol | FL/FR |
| | phenyl acetic acid | FL/FR |
| | tetrahydrolinalool | FL/FR |
| | tuberose absolute (from concrete) | FL/FR |
| | tuberose absolute (from pommade) | FL/FR |
| fruity |
| | allyl cyclohexyl propionate | FL/FR |
| iso | amyl benzoate | FL/FR |
| iso | amyl isobutyrate | FL/FR |
| para- | anisyl alcohol | FL/FR |
| | benzyl methyl ether | FL/FR |
| | diethyl succinate | FL/FR |
| | dimethyl succinate | FL/FR |
| | ethyl methyl-para-tolyl glycidate | FL/FR |
| 2- | hexen-1-ol | FL/FR |
| | hexyl acetate | FL/FR |
| 2- | phenyl propionaldehyde dimethyl acetal | FL/FR |
| | rose butanoate | FL/FR |
| | styralyl acetate | FL/FR |
| iso | valeraldehyde propylene glycol acetal | FL/FR |
| green |
| | acetaldehyde ethyl phenethyl acetal | FL/FR |
| iso | amyl isovalerate | FL/FR |
| iso | amyl salicylate | FL/FR |
| | chrysanthemum oxide | FL/FR |
| | cinnamyl alcohol | FL/FR |
| | cucumber distillates | FL |
| | cuminyl acetaldehyde | FL/FR |
| | dihydromyrcenol | FL/FR |
| 2,5- | dimethyl-3,6-dihexyl pyrazine | |
| | diphenyl methane | FL/FR |
| | diphenyl oxide | FL/FR |
| | geranyl acetate | FL/FR |
| | green ether | FL/FR |
| | heptyl acetate | FL/FR |
| (Z)-3- | hexen-1-yl salicylate | FL/FR |
| | hexyl tiglate | FL/FR |
| (Z)- | leaf acetal | FL/FR |
| 3,5- | lutidine | FL |
| | melon nonenoate | FL/FR |
| | methyl 2-undecynoate | FL |
| para- | methyl hydratropaldehyde | FL/FR |
| | methyl octine carbonate | FL/FR |
| | nerolidol | FL/FR |
| (E)-2- | nonen-1-ol | FL/FR |
| (Z)-6- | nonen-1-yl acetate | FL/FR |
| | oakmoss absolute | FL/FR |
| | octanal dimethyl acetal | FL/FR |
| (E)-2- | octen-1-yl acetate | FL/FR |
| | papaya isobutyrate | FL/FR |
| | phenethyl formate | FL/FR |
| | phenyl acetaldehyde diisobutyl acetal | FL/FR |
| | phenyl acetaldehyde dimethyl acetal | FL/FR |
| 2- | phenyl propionaldehyde | FL/FR |
| 3- | phenyl propionaldehyde | FL/FR |
| 2- | phenyl propyl alcohol | FL/FR |
| | violet leaf absolute | FL/FR |
| herbal |
| | clary sage absolute | FL/FR |
| | clary sage oil france | FL/FR |
| | coriander seed oil | FL/FR |
| | daucus carota fruit oil | FL/FR |
| | lavender absolute bulgaria | FL/FR |
| | ocimum basilicum herb oil | FL/FR |
| honey |
| | phenyl acetaldehyde | FL/FR |
| medicinal |
| | dimethyl benzyl carbinol | FL/FR |
| | phenethyl salicylate | FL/FR |
| melon |
| | hydroxycitronellal diethyl acetal | FL/FR |
| minty |
| | spearmint absolute | FL/FR |
| naphthyl |
| para- | methyl anisole | FL/FR |
| oily |
| | olive oil absolute | FL/FR |
| phenolic |
| para- | cresyl phenyl acetate | FL/FR |
| pine |
| beta- | pinene | FL/FR |
| ripe |
| (E)-4- | decenal | FL/FR |
| solvent |
| 1- | heptanol | FL/FR |
| spicy |
| | cinnamyl formate | FL/FR |
| | clove bud oil | FL/FR |
| | cuminaldehyde | FL/FR |
| black | currant bud absolute | FL/FR |
| | eugenol | FL/FR |
| | methyl cinnamate | FL/FR |
| 3- | phenyl propyl alcohol | FL/FR |
| | pimenta acris leaf oil | FL/FR |
| tropical |
| alpha- | amyl cinnamaldehyde | FL/FR |
| vanilla |
| | ethyl vanillin | FL/FR |
| | vanilla bean absolute (vanilla planifolia) | FL/FR |
| | vanillyl acetate | FL/FR |
| waxy |
| iso | amyl butyrate | FL/FR |
| | decanol | FL/FR |
| | ethyl laurate | FL/FR |
| alpha- | hexyl cinnamaldehyde | FL/FR |
| | mimosa absolute france | FL/FR |
| woody |
| | amyris wood oil | FL/FR |
| | guaiacwood oil | FL/FR |
| | vetiver oil haiti | FL/FR |
| |
Potential Uses:
Occurrence (nature, food, other): noteSynonyms:
| | benzene, 1,4-dimethoxy- | | | di methyl hydroquinone | | 1,4- | dimethoxy benzene | | 1,4- | dimethoxy-benzene | | 1,4- | dimethoxybenzene | | 4- | dimethoxybenzene | | p- | dimethoxybenzene | | para- | dimethoxybenzene | | 1,4- | dimethoxybenzol | | | dimethyl ether hydroquinone | | | dimethyl hydroquinone | | p- | dimethyl hydroquinone | | | dimethyl hydroquinone ether | | | dimethylhydroquinone | | p- | dimethylhydroquinone | | | hydroquinone dimethyl ether | | | hydrquinone dimethyl ether | | p- | methoxyanisole | | para- | methoxyanisole | | | methyl p-methoxyphenyl ether | | | methyl para-methoxyphenyl ether | | | quinol dimethyl ether |
Articles:
| PubMed: | Theoretic calculation for understanding the oxidation process of 1,4-dimethoxybenzene-based compounds as redox shuttles for overcharge protection of lithium ion batteries. |
| PubMed: | Intramolecular electronic couplings in class II/III organic mixed-valence systems of bis(1,4-dimethoxybenzene). |
| PubMed: | Ozonolysis of lignin models in aqueous solution: anisole, 1,2-dimethoxybenzene, 1,4-dimethoxybenzene, and 1,3,5-trimethoxybenzene. |
| PubMed: | Perception of floral volatiles involved in host-plant finding behaviour: comparison of a bee specialist and generalist. |
| PubMed: | Highly stable high performance liquid chromatography stationary phase based on direct chemical modification of organic bridges in hybrid silica. |
| PubMed: | 1,4-Dimethoxybenzene, a floral scent compound in willows that attracts an oligolectic bee. |
| PubMed: | 2-Chloro-1,4-dimethoxybenzene as a mediator of lignin peroxidase catalyzed oxidations. |
| PubMed: | 2-Chloro-1,4-dimethoxybenzene cation radical: formation and role in the lignin peroxidase oxidation of anisyl alcohol. |
| PubMed: | Size-selected gold clusters on porous titania as the most "gold-efficient" heterogeneous catalysts. |
| PubMed: | 2-chloro-1,4-dimethoxybenzene as a novel catalytic cofactor for oxidation of anisyl alcohol by lignin peroxidase. |
| PubMed: | The de novo production of drosophilin A (tetrachloro-4-methoxyphenol) and drosophilin A methyl ether (tetrachloro-1,4-dimethoxybenzene) by ligninolytic basidiomycetes. |
| PubMed: | [Antibacterial activity of secondary metabolites from Aspergillus awamori F12 isolated from rhizospheric soil of Rhizophora stylosa Griff]. |
| PubMed: | Aerobic oxidative coupling of arenes and olefins through a biomimetic approach. |
| PubMed: | Simultaneous determination of charge number and diffusion coefficient of 2-chloro-1,4-dimethoxybenzene in acetonitrile using potential-step chronoamperometry. |
| PubMed: | Reversible photochemically gated transformation of a hemicarcerand to a carcerand. |
| PubMed: | Degradation and transformation products of acetaminophen in soil. |
| PubMed: | Ecological relationship between floral thermogenesis and pollination in Nelumbo lutea (Nelumbonaceae). |
| PubMed: | Hydrogen-bonded inclusion compounds with reversed polarity: anionic metal-complexes and cationic organic linkers. |
| PubMed: | Quantitative preparative gas chromatography of caffeine with nuclear magnetic resonance spectroscopy. |
| PubMed: | Halogen-substituted (C-β-D-glucopyranosyl)-hydroquinone regioisomers: synthesis, enzymatic evaluation and their binding to glycogen phosphorylase. |
| PubMed: | Synthesis and X-ray crystal structure of a difunctionalized pillar[5]arene at A1/B2 positions by in situ cyclization and deprotection. |
| PubMed: | Epimeric monosaccharide-quinone hybrids on gold electrodes toward the electrochemical probing of specific carbohydrate-protein recognitions. |
| PubMed: | Regioselectivity in the nitration of dialkoxybenzenes. |
| PubMed: | Transcriptional response of zebrafish embryos exposed to neurotoxic compounds reveals a muscle activity dependent hspb11 expression. |
| PubMed: | Pairwise substitution effects, inter- and intramolecular hydrogen bonds in methoxyphenols and dimethoxybenzenes. Thermochemistry, calorimetry, and first-principles calculations. |
| PubMed: | Nitration of veratryl alcohol by lignin peroxidase and tetranitromethane. |
| PubMed: | Oxidation of dimethoxylated aromatic compounds by lignin peroxidase from Phanerochaete chrysosporium. |
| PubMed: | Deprotonative metalation of aromatic compounds by using an amino-based lithium cuprate. |
| PubMed: | Crystallographic, kinetic, and spectroscopic study of the first ligninolytic peroxidase presenting a catalytic tyrosine. |
| PubMed: | Guest-induced assembly of tetracarboxyl-cavitand and tetra(3-pyridyl)-cavitand into a heterodimeric capsule via hydrogen bonds and CH-halogen and/or CH-pi interaction: control of the orientation of the encapsulated guest. |
| PubMed: | Escherichia coli expression and in vitro activation of a unique ligninolytic peroxidase that has a catalytic tyrosine residue. |
| PubMed: | Analysis of biogenic volatile organic compounds in zucchini flowers: identification of scent sources. |
| PubMed: | Reactions within p-difluorobenzene/methanol heterocluster ions: a detailed experimental and theoretical investigation. |
| PubMed: | Degradation of 2,4-dichlorophenol by the lignin-degrading fungus Phanerochaete chrysosporium. |
| PubMed: | Application of microscale-preparative multidimensional gas chromatography with nuclear magnetic resonance spectroscopy for identification of pure methylnaphthalenes from crude oils. |
| PubMed: | Degradation of various alkyl ethers by alkyl ether-degrading Actinobacteria isolated from activated sludge of a mixed wastewater treatment. |
| PubMed: | Induction of forestomach lesions by butylhydroxyanisole and structurally related substances. |
| PubMed: | Inter- and intraspecific variation in floral scent in the genus salix and its implication for pollination. |
| PubMed: | para-Bridged symmetrical pillar[5]arenes: their Lewis acid catalyzed synthesis and host-guest property. |
| PubMed: | Photoarylation/alkylation of bromonaphthols. |
| PubMed: | Variability in floral scent in rewarding and deceptive orchids: the signature of pollinator-imposed selection? |
| PubMed: | Photosensitized oxidation of sulfides: discriminating between the singlet-oxygen mechanism and electron transfer involving superoxide anion or molecular oxygen. |
| PubMed: | Anodic oxidation of mono- and disubstituted 1,4-dimethoxybenzenes. |
| PubMed: | One-pot synthesis of C-glycosylic compounds (C-glycosides) from D-glucal, p-tolylsulfenyl chloride and aromatic/heteroaromatic compounds in the presence of Lewis acids. |
| PubMed: | Photochemistry of methoxyhydroquinone and methoxy-p-benzoquinone in solution related to the photoyellowing of the lignocellulosics. |
| PubMed: | Cis-Trans Isomerization and Oxidation of Radical Cations of Stilbene Derivatives. |
| PubMed: | Rate parameter changes by added albumin in the microsomal oxidative demethylation of deuteriated and non-deuteriated 4-methoxyanisole. |
| PubMed: | [Quantitative analysis of tripchlorolide in pharmaceutical preparation by RP-HPLC]. |
| PubMed: | Mechanism of antitumoral activity of catechols in culture. |
| PubMed: | The ligninase of Phanerochaete chrysosporium generates cation radicals from methoxybenzenes. |
| PubMed: | Synthesis of new phytogrowth-inhibitory substituted aryl-p-benzoquinones. |
| PubMed: | Selective monoiodination of aromatic compounds with electrochemically generated I+ using micromixing. |
| PubMed: | Microbial anaerobic demethylation and dechlorination of chlorinated hydroquinone metabolites synthesized by basidiomycete fungi. |
| PubMed: | First synthesis of a polysaccharide-supported lignin model compound and study of its oxidation promoted by lignin peroxidase. |
| PubMed: | Role of face-to-face and edge-to-face aromatic interactions in the inclusion complexation of cyclobis(paraquat-p-phenylene): a theoretical study. |
| PubMed: | [Studies on the chemical constituents of chloroform extract of Dracaena cochinchinensis]. |
| PubMed: | Facile oxidation of fused 1,4-dimethoxybenzenes to 1,4-quinones using NBS: fine-tuned control over bromination and oxidation reactions. |
| PubMed: | Volatile methoxybenzene compounds in grains with off-odors. |
| PubMed: | Effects of BHA and related phenols on the forestomach of rats. |
| PubMed: | Decrease in glucose oxidation in isolated brown fat cells from rats due to tropolone and dimethoxybenzene. |
| PubMed: | Reactions of N-methyl-N-(4-biphenylyl)nitrenium ion with electron-rich arenes: laser flash photolysis and product studies. |
| PubMed: | Synthesis of indan-based unusual alpha-amino acid derivatives under phase-transfer catalysis conditions. |
| PubMed: | Characterization of a novel manganese peroxidase-lignin peroxidase hybrid isozyme produced by Bjerkandera species strain BOS55 in the absence of manganese. |
| PubMed: | On the mechanism of inhibition of the veratryl alcohol oxidase activity of lignin peroxidase H2 by EDTA. |
| PubMed: | Are free radicals involved in tumor promotion? |
| PubMed: | Metallocyclic receptors with Re(I)/Os(II)-based moieties: molecular photophysics and selective molecular sensing. |
| PubMed: | Iodide as the mediator for the reductive reactions of peroxidases. |
| PubMed: | Transformation of chlorinated phenolic compounds in the genusRhodococcus. |
| PubMed: | Protective effects of butylated hydroxyanisole and its analogs on the lung toxicity of butylated hydroxytoluene in mice. |
| PubMed: | Inhibition by 2(3)-tert-butyl-4-hydroxyanisole and other antioxidants of epidermal ornithine decarboxylase activity induced by 12-O-tetradecanoylphorbol-13-acetate. |
|
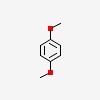 3D/inchi
3D/inchi


















